Mechanosensitive Ca(2+) permeant cation channels in human prostate tumor cells
- PMID: 22874798
- PMCID: PMC3508908
- DOI: 10.4161/chan.21063
Mechanosensitive Ca(2+) permeant cation channels in human prostate tumor cells
Abstract
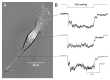
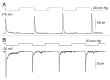
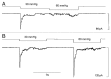
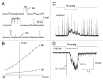
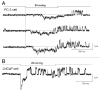
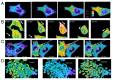
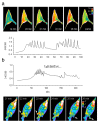
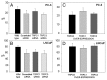
The acquisition of cell motility plays a critical role in the spread of prostate cancer (PC), therefore, identifying a sensitive step that regulates PC cell migration should provide a promising target to block PC metastasis. Here, we report that a mechanosensitive Ca(2+)-permeable cation channel (MscCa) is expressed in the highly migratory/invasive human PC cell line, PC-3 and that inhibition of MscCa by Gd(3+) or GsMTx-4 blocks PC-3 cell migration and associated elevations in [Ca(2+)](i). Genetic suppression or overexpression of specific members of the canonical transient receptor potential Ca(2+) channel family (TRPC1 and TRPC3) also inhibit PC-3 cell migration, but they do so by mechanisms other that altering MscCa activity. Although LNCaP cells are nonmigratory, they also express relatively large MscCa currents, indicating that MscCa expression alone cannot confer motility on PC cells. MscCa in both cell lines show similar conductance and ion selectivity and both are functionally coupled via Ca(2+) influx to a small Ca(2+)-activated K(+) channel. However, MscCa in PC-3 and LNCaP cell patches show markedly different gating dynamics--while PC-3 cells typically express a sustained, non-inactivating MscCa current, LNCaP cells express a mechanically-fragile, rapidly inactivating MscCa current. Moreover, mechanical forces applied to the patch, can induce an irreversible transition from the transient to the sustained MscCa gating mode. Given that cancer cells experience increasing compressive and shear forces within a growing tumor, a similar shift in channel gating in situ would have significant effects on Ca(2+) signaling that may play a role in tumor progression.
Figures







Similar articles
-
TRPC1 forms the stretch-activated cation channel in vertebrate cells.Nat Cell Biol. 2005 Feb;7(2):179-85. doi: 10.1038/ncb1218. Epub 2005 Jan 23. Nat Cell Biol. 2005. PMID: 15665854
-
TRPC1 channels regulate directionality of migrating cells.Pflugers Arch. 2008 Nov;457(2):475-84. doi: 10.1007/s00424-008-0515-4. Epub 2008 Apr 30. Pflugers Arch. 2008. PMID: 18542994
-
Calcium store contents control the expression of TRPC1, TRPC3 and TRPV6 proteins in LNCaP prostate cancer cell line.Cell Calcium. 2006 May;39(5):401-15. doi: 10.1016/j.ceca.2006.01.003. Epub 2006 Mar 9. Cell Calcium. 2006. PMID: 16529812
-
Transient Receptor Potential Canonical (TRPC) Channels as Modulators of Migration and Invasion.Int J Mol Sci. 2020 Mar 3;21(5):1739. doi: 10.3390/ijms21051739. Int J Mol Sci. 2020. PMID: 32138386 Free PMC article. Review.
-
Transient receptor potential channels in endothelium: solving the calcium entry puzzle?Endothelium. 2003;10(1):5-15. doi: 10.1080/10623320303356. Endothelium. 2003. PMID: 12699072 Review.
Cited by
-
Mechanosensitive ion channels: an evolutionary and scientific tour de force in mechanobiology.Channels (Austin). 2012 Jul-Aug;6(4):211-3. doi: 10.4161/chan.22047. Epub 2012 Jul 1. Channels (Austin). 2012. PMID: 22940794 Free PMC article. No abstract available.
-
Feeling Stress: The Mechanics of Cancer Progression and Aggression.Front Cell Dev Biol. 2018 Feb 28;6:17. doi: 10.3389/fcell.2018.00017. eCollection 2018. Front Cell Dev Biol. 2018. PMID: 29541636 Free PMC article. Review.
-
Biophysical interactions between components of the tumor microenvironment promote metastasis.Biophys Rev. 2021 Jun 4;13(3):339-357. doi: 10.1007/s12551-021-00811-y. eCollection 2021 Jun. Biophys Rev. 2021. PMID: 34168685 Free PMC article. Review.
-
Piezo1, the new actor in cell volume regulation.Pflugers Arch. 2024 Jul;476(7):1023-1039. doi: 10.1007/s00424-024-02951-y. Epub 2024 Apr 6. Pflugers Arch. 2024. PMID: 38581527 Free PMC article. Review.
-
Docetaxel modulates the delayed rectifier potassium current (IK) and ATP-sensitive potassium current (IKATP) in human breast cancer cells.J Membr Biol. 2015 Apr;248(2):197-204. doi: 10.1007/s00232-014-9757-6. Epub 2014 Nov 25. J Membr Biol. 2015. PMID: 25421218
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
