The dynamics of protein-protein interactions between domains of MscL at the cytoplasmic-lipid interface
- PMID: 22874845
- PMCID: PMC3508904
- DOI: 10.4161/chan.20756
The dynamics of protein-protein interactions between domains of MscL at the cytoplasmic-lipid interface
Abstract
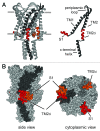
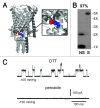
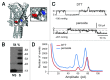
The bacterial mechanosensitive channel of large conductance, MscL, is one of the best characterized mechanosensitive channels serving as a paradigm for how proteins can sense and transduce mechanical forces. The physiological role of MscL is that of an emergency release valve that opens a large pore upon a sudden drop in the osmolarity of the environment. A crystal structure of a closed state of MscL shows it as a homopentamer, with each subunit consisting of two transmembrane domains (TM). There is consensus that the TM helices move in an iris like manner tilting in the plane of the membrane while gating. An N-terminal amphipathic helix that lies along the cytoplasmic membrane (S1), and the portion of TM2 near the cytoplasmic interface (TM2(ci)), are relatively close in the crystal structure, yet predicted to be dynamic upon gating. Here we determine how these two regions interact in the channel complex, and study how these interactions change as the channel opens. We have screened 143 double-cysteine mutants of E. coli MscL for their efficiency in disulfide bridging and generated a map of protein-protein interactions between these two regions. Interesting candidates have been further studied by patch clamp and show differences in channel activity under different redox potentials; the results suggest a model for the dynamics of these two domains during MscL gating.
Figures

Similar articles
-
On the structure of the N-terminal domain of the MscL channel: helical bundle or membrane interface.Biophys J. 2008 Sep;95(5):2283-91. doi: 10.1529/biophysj.107.127423. Epub 2008 May 30. Biophys J. 2008. PMID: 18515388 Free PMC article.
-
Disulfide trapping the mechanosensitive channel MscL into a gating-transition state.Biophys J. 2007 Feb 15;92(4):1224-32. doi: 10.1529/biophysj.106.090316. Epub 2006 Nov 17. Biophys J. 2007. PMID: 17114217 Free PMC article.
-
The gating mechanism of the bacterial mechanosensitive channel MscL revealed by molecular dynamics simulations: from tension sensing to channel opening.Channels (Austin). 2012 Jul-Aug;6(4):317-31. doi: 10.4161/chan.21895. Channels (Austin). 2012. PMID: 23146938 Free PMC article.
-
Two families of mechanosensitive channel proteins.Microbiol Mol Biol Rev. 2003 Mar;67(1):66-85, table of contents. doi: 10.1128/MMBR.67.1.66-85.2003. Microbiol Mol Biol Rev. 2003. PMID: 12626684 Free PMC article. Review.
-
Emerging issues of connexin channels: biophysics fills the gap.Q Rev Biophys. 2001 Aug;34(3):325-472. doi: 10.1017/s0033583501003705. Q Rev Biophys. 2001. PMID: 11838236 Review.
Cited by
-
Novel MscL agonists that allow multiple antibiotics cytoplasmic access activate the channel through a common binding site.PLoS One. 2020 Jan 24;15(1):e0228153. doi: 10.1371/journal.pone.0228153. eCollection 2020. PLoS One. 2020. PMID: 31978161 Free PMC article.
-
Novel compounds that specifically bind and modulate MscL: insights into channel gating mechanisms.FASEB J. 2019 Mar;33(3):3180-3189. doi: 10.1096/fj.201801628R. Epub 2018 Oct 25. FASEB J. 2019. PMID: 30359098 Free PMC article.
-
Life with Bacterial Mechanosensitive Channels, from Discovery to Physiology to Pharmacological Target.Microbiol Mol Biol Rev. 2020 Jan 15;84(1):e00055-19. doi: 10.1128/MMBR.00055-19. Print 2020 Feb 19. Microbiol Mol Biol Rev. 2020. PMID: 31941768 Free PMC article. Review.
-
Scanning MscL Channels with Targeted Post-Translational Modifications for Functional Alterations.PLoS One. 2015 Sep 14;10(9):e0137994. doi: 10.1371/journal.pone.0137994. eCollection 2015. PLoS One. 2015. PMID: 26368283 Free PMC article.
-
Mechanical coupling of the multiple structural elements of the large-conductance mechanosensitive channel during expansion.Proc Natl Acad Sci U S A. 2015 Aug 25;112(34):10726-31. doi: 10.1073/pnas.1503202112. Epub 2015 Aug 10. Proc Natl Acad Sci U S A. 2015. PMID: 26261325 Free PMC article.
References
-
- Blount P, Iscla I, Moe PC, Li Y. MscL: The Bacterial Mechanosensitive Channel of Large Conductance. In: Hamill O, ed. Current Topics in Membranes: Academic Press, 2007:201-33.
-
- Levina N, Tötemeyer S, Stokes NR, Louis P, Jones MA, Booth IR. Protection of Escherichia coli cells against extreme turgor by activation of MscS and MscL mechanosensitive channels: identification of genes required for MscS activity. EMBO J. 1999;18:1730–7. doi: 10.1093/emboj/18.7.1730. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
