Atonal and EGFR signalling orchestrate rok- and Drak-dependent adherens junction remodelling during ommatidia morphogenesis
- PMID: 22874916
- PMCID: PMC3424046
- DOI: 10.1242/dev.080762
Atonal and EGFR signalling orchestrate rok- and Drak-dependent adherens junction remodelling during ommatidia morphogenesis
Abstract
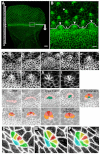
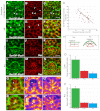
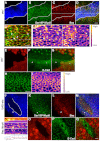
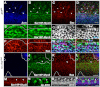
Morphogenesis of epithelial tissues relies on the interplay between cell division, differentiation and regulated changes in cell shape, intercalation and sorting. These processes are often studied individually in relatively simple epithelia that lack the complexity found during organogenesis when these processes might all coexist simultaneously. To address this issue, we are making use of the developing fly retinal neuroepithelium. Retinal morphogenesis relies on a coordinated sequence of interdependent morphogenetic events that includes apical cell constriction, localized alignment of groups of cells and ommatidia morphogenesis coupled to neurogenesis. Here, we use live imaging to document the sequence of adherens junction (AJ) remodelling events required to generate the fly ommatidium. In this context, we demonstrate that the kinases Rok and Drak function redundantly during Myosin II-dependent cell constriction, subsequent multicellular alignment and AJ remodelling. In addition, we show that early multicellular patterning characterized by cell alignment is promoted by the conserved transcription factor Atonal (Ato). Further ommatidium patterning requires the epidermal growth factor receptor (EGFR) signalling pathway, which transcriptionally governs rok- and Drak-dependent AJ remodelling while also promoting neurogenesis. In conclusion, our work reveals an important role for Drak in regulating AJ remodelling during retinal morphogenesis. It also sheds new light on the interplay between Ato, EGFR-dependent transcription and AJ remodelling in a system in which neurogenesis is coupled with cell shape changes and regulated steps of cell intercalation.
Figures


Similar articles
-
Overlapping roles of Drosophila Drak and Rok kinases in epithelial tissue morphogenesis.Mol Biol Cell. 2010 Aug 15;21(16):2869-79. doi: 10.1091/mbc.E10-04-0328. Epub 2010 Jun 23. Mol Biol Cell. 2010. PMID: 20573980 Free PMC article.
-
The vav oncogene antagonises EGFR signalling and regulates adherens junction dynamics during Drosophila eye development.Development. 2015 Apr 15;142(8):1492-501. doi: 10.1242/dev.110585. Epub 2015 Mar 26. Development. 2015. PMID: 25813543
-
Epithelial cell adhesion in the developing Drosophila retina is regulated by Atonal and the EGF receptor pathway.Dev Biol. 2006 Dec 15;300(2):710-21. doi: 10.1016/j.ydbio.2006.08.003. Epub 2006 Aug 9. Dev Biol. 2006. PMID: 16963016
-
Hexagonal patterning of the Drosophila eye.Dev Biol. 2021 Oct;478:173-182. doi: 10.1016/j.ydbio.2021.07.004. Epub 2021 Jul 8. Dev Biol. 2021. PMID: 34245727 Free PMC article. Review.
-
Regulatory mechanisms of EGFR signalling during Drosophila eye development.Cell Mol Life Sci. 2016 May;73(9):1825-43. doi: 10.1007/s00018-016-2153-x. Epub 2016 Mar 2. Cell Mol Life Sci. 2016. PMID: 26935860 Free PMC article. Review.
Cited by
-
Drak/STK17A Drives Neoplastic Glial Proliferation through Modulation of MRLC Signaling.Cancer Res. 2019 Mar 15;79(6):1085-1097. doi: 10.1158/0008-5472.CAN-18-0482. Epub 2018 Dec 10. Cancer Res. 2019. PMID: 30530503 Free PMC article.
-
Spatiotemporal control of epithelial remodeling by regulated myosin phosphorylation.Proc Natl Acad Sci U S A. 2014 Aug 12;111(32):11732-7. doi: 10.1073/pnas.1400520111. Epub 2014 Jul 28. Proc Natl Acad Sci U S A. 2014. PMID: 25071215 Free PMC article.
-
An improved organ explant culture method reveals stem cell lineage dynamics in the adult Drosophila intestine.Elife. 2022 Aug 25;11:e76010. doi: 10.7554/eLife.76010. Elife. 2022. PMID: 36005292 Free PMC article.
-
Super-resolution imaging in whole cells and tissues via DNA-PAINT on a spinning disk confocal with optical photon reassignment.Nat Commun. 2025 May 29;16(1):4991. doi: 10.1038/s41467-025-60263-w. Nat Commun. 2025. PMID: 40442066 Free PMC article.
-
MRCK: a master regulator of tissue remodeling or another 'ROCK' in the epithelial block?Tissue Barriers. 2021 Jul 3;9(3):1916380. doi: 10.1080/21688370.2021.1916380. Epub 2021 May 19. Tissue Barriers. 2021. PMID: 34008470 Free PMC article. Review.
References
-
- Abramoff M. D., Magelhaes P. J., Ram S. J. (2004). Image processing with Image J. Biophotonics International 11, 36–42
-
- Baonza A., Casci T., Freeman M. (2001). A primary role for the epidermal growth factor receptor in ommatidial spacing in the Drosophila eye. Curr. Biol. 11, 396–404 - PubMed
-
- Bertet C., Sulak L., Lecuit T. (2004). Myosin-dependent junction remodelling controls planar cell intercalation and axis elongation. Nature 429, 667–671 - PubMed
-
- Blankenship J. T., Backovic S. T., Sanny J. S., Weitz O., Zallen J. A. (2006). Multicellular rosette formation links planar cell polarity to tissue morphogenesis. Dev. Cell 11, 459–470 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

