Supramolecular hydrogel of kanamycin selectively sequesters 16S rRNA
- PMID: 22875345
- PMCID: PMC3472799
- DOI: 10.1039/c2cc34935c
Supramolecular hydrogel of kanamycin selectively sequesters 16S rRNA
Abstract
As the first example of hydrogelator derived from aminoglycoside antibiotics, the hydrogel of kanamycin indicates that the hydrogel of aminoglycosides preserve the specific interaction with their macromolecular targets (e.g., 16S rRNA), thus illustrating a simple approach to explore and identify possible biological targets of supramolecular nanofibers/hydrogels.
Figures

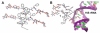


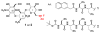
Similar articles
-
Docking of cationic antibiotics to negatively charged pockets in RNA folds.J Med Chem. 1999 Apr 8;42(7):1250-61. doi: 10.1021/jm981108g. J Med Chem. 1999. PMID: 10197968
-
Interactions of aminoglycoside antibiotics with rRNA.Biochem Soc Trans. 2016 Aug 15;44(4):987-93. doi: 10.1042/BST20160087. Biochem Soc Trans. 2016. PMID: 27528743 Review.
-
Fluorescence-based approach for detecting and characterizing antibiotic-induced conformational changes in ribosomal RNA: comparing aminoglycoside binding to prokaryotic and eukaryotic ribosomal RNA sequences.J Am Chem Soc. 2004 Mar 24;126(11):3447-53. doi: 10.1021/ja030568i. J Am Chem Soc. 2004. PMID: 15025471
-
Enhanced binding of aminoglycoside dimers to a "dimerized" A-site 16S rRNA construct.Bioorg Med Chem Lett. 2000 Jul 17;10(14):1593-5. doi: 10.1016/s0960-894x(00)00299-7. Bioorg Med Chem Lett. 2000. PMID: 10915059
-
The binding of antibiotics to RNA.Prog Biophys Mol Biol. 1997;67(2-3):141-54. doi: 10.1016/s0079-6107(97)00011-4. Prog Biophys Mol Biol. 1997. PMID: 9446933 Review. No abstract available.
Cited by
-
Supramolecular Hydrogelators and Hydrogels: From Soft Matter to Molecular Biomaterials.Chem Rev. 2015 Dec 23;115(24):13165-307. doi: 10.1021/acs.chemrev.5b00299. Epub 2015 Dec 8. Chem Rev. 2015. PMID: 26646318 Free PMC article. Review.
-
Self-delivery multifunctional anti-HIV hydrogels for sustained release.Adv Healthc Mater. 2013 Dec;2(12):1586-90. doi: 10.1002/adhm.201300041. Epub 2013 Apr 25. Adv Healthc Mater. 2013. PMID: 23616384 Free PMC article.
-
Supramolecular hydrogels made of basic biological building blocks.Chem Asian J. 2014 Jun;9(6):1446-72. doi: 10.1002/asia.201301693. Epub 2014 Mar 12. Chem Asian J. 2014. PMID: 24623474 Free PMC article. Review.
-
Dephosphorylation of D-peptide derivatives to form biofunctional, supramolecular nanofibers/hydrogels and their potential applications for intracellular imaging and intratumoral chemotherapy.J Am Chem Soc. 2013 Jul 3;135(26):9907-14. doi: 10.1021/ja404215g. Epub 2013 Jun 21. J Am Chem Soc. 2013. PMID: 23742714 Free PMC article.
-
Building Nanostructures with Drugs.Nano Today. 2016 Feb;11(1):13-30. doi: 10.1016/j.nantod.2015.11.003. Nano Today. 2016. PMID: 27066106 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

