The role of the subthalamic nucleus in L-DOPA induced dyskinesia in 6-hydroxydopamine lesioned rats
- PMID: 22880070
- PMCID: PMC3412805
- DOI: 10.1371/journal.pone.0042652
The role of the subthalamic nucleus in L-DOPA induced dyskinesia in 6-hydroxydopamine lesioned rats
Abstract
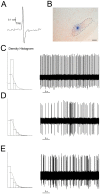
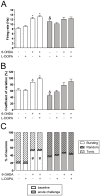
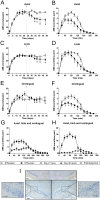
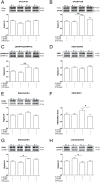
L-DOPA is the most effective treatment for Parkinson's disease (PD), but prolonged use leads to disabling motor complications including dyskinesia. Strong evidence supports a role of the subthalamic nucleus (STN) in the pathophysiology of PD whereas its role in dyskinesia is a matter of controversy. Here, we investigated the involvement of STN in dyskinesia, using single-unit extracellular recording, behavioural and molecular approaches in hemi-parkinsonian rats rendered dyskinetic by chronic L-DOPA administration. Our results show that chronic L-DOPA treatment does not modify the abnormal STN activity induced by the 6-hydroxydopamine lesion of the nigrostriatal pathway in this model. Likewise, we observed a loss of STN responsiveness to a single L-DOPA dose both in lesioned and sham animals that received daily L-DOPA treatment. We did not find any correlation between the abnormal involuntary movement (AIM) scores and the electrophysiological parameters of STN neurons recorded 24 h or 20-120 min after the last L-DOPA injection, except for the axial subscores. Nonetheless, unilateral chemical ablation of the STN with ibotenic acid resulted in a reduction in global AIM scores and peak-severity of dyskinesia. In addition, STN lesion decreased the anti-dyskinetogenic effect of buspirone in a reciprocal manner. Striatal protein expression was altered in dyskinetic animals with increases in ΔFosB, phosphoDARPP-32, dopamine receptor (DR) D3 and DRD2/DRD1 ratio. The STN lesion attenuated the striatal molecular changes and normalized the DRD2/DRD1 ratio. Taken together, our results show that the STN plays a role, if modest, in the physiopathology of dyskinesias.
Conflict of interest statement
Figures



Similar articles
-
Chronic L-DOPA administration increases the firing rate but does not reverse enhanced slow frequency oscillatory activity and synchronization in substantia nigra pars reticulata neurons from 6-hydroxydopamine-lesioned rats.Neurobiol Dis. 2016 May;89:88-100. doi: 10.1016/j.nbd.2016.02.003. Epub 2016 Feb 4. Neurobiol Dis. 2016. PMID: 26852950
-
Buspirone requires the intact nigrostriatal pathway to reduce the activity of the subthalamic nucleus via 5-HT1A receptors.Exp Neurol. 2016 Mar;277:35-45. doi: 10.1016/j.expneurol.2015.12.005. Epub 2015 Dec 11. Exp Neurol. 2016. PMID: 26687972
-
Acute L-DOPA administration reverses changes in firing pattern and low frequency oscillatory activity in the entopeduncular nucleus from long term L-DOPA treated 6-OHDA-lesioned rats.Exp Neurol. 2019 Dec;322:113036. doi: 10.1016/j.expneurol.2019.113036. Epub 2019 Aug 16. Exp Neurol. 2019. PMID: 31425688
-
Dyskinesias and the subthalamic nucleus.Ann Neurol. 2000 Apr;47(4 Suppl 1):S189-92. Ann Neurol. 2000. PMID: 10762147 Review.
-
The interhemispheric connections of the striatum: Implications for Parkinson's disease and drug-induced dyskinesias.Brain Res Bull. 2012 Jan 4;87(1):1-9. doi: 10.1016/j.brainresbull.2011.09.013. Epub 2011 Sep 24. Brain Res Bull. 2012. PMID: 21963946 Free PMC article. Review.
Cited by
-
Motor cortico-nigral and cortico-entopeduncular information transmission and its modulation by buspirone in control and after dopaminergic denervation.Front Pharmacol. 2022 Aug 30;13:953652. doi: 10.3389/fphar.2022.953652. eCollection 2022. Front Pharmacol. 2022. PMID: 36133803 Free PMC article.
-
6-Hydroxydopamine lesion and levodopa treatment modify the effect of buspirone in the substantia nigra pars reticulata.Br J Pharmacol. 2020 Sep;177(17):3957-3974. doi: 10.1111/bph.15145. Epub 2020 Jul 6. Br J Pharmacol. 2020. PMID: 32464686 Free PMC article.
-
Neurobiological and Pharmacological Perspectives of D3 Receptors in Parkinson's Disease.Biomolecules. 2022 Feb 1;12(2):243. doi: 10.3390/biom12020243. Biomolecules. 2022. PMID: 35204744 Free PMC article. Review.
-
Cortical Control of Subthalamic Neuronal Activity through the Hyperdirect and Indirect Pathways in Monkeys.J Neurosci. 2020 Sep 23;40(39):7451-7463. doi: 10.1523/JNEUROSCI.0772-20.2020. Epub 2020 Aug 26. J Neurosci. 2020. PMID: 32847963 Free PMC article.
-
Morphological Changes in a Severe Model of Parkinson's Disease and Its Suitability to Test the Therapeutic Effects of Microencapsulated Neurotrophic Factors.Mol Neurobiol. 2017 Dec;54(10):7722-7735. doi: 10.1007/s12035-016-0244-1. Epub 2016 Nov 14. Mol Neurobiol. 2017. PMID: 27844282
References
-
- Kish SJ, Shannak K, Hornykiewicz O (1988) Uneven pattern of dopamine loss in the striatum of patients with idiopathic Parkinson's disease. Pathophysiologic and clinical implications. N Engl J Med 318: 876–880. - PubMed
-
- Ahlskog JE, Muenter MD (2001) Frequency of levodopa-related dyskinesias and motor fluctuations as estimated from the cumulative literature. Mov Disord 16: 448–458. - PubMed
-
- Obeso JA, Olanow CW, Nutt JG (2000) Levodopa motor complications in Parkinson's disease. Trends Neurosci 23: S2–S7. - PubMed
-
- Cesaro P, Damier P, Nguyen JP, Palfi S, Ollat H (2003) La maladie de Parkinson.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

