Intragraft CD11b(+) IDO(+) cells mediate cardiac allograft tolerance by ECDI-fixed donor splenocyte infusions
- PMID: 22883222
- PMCID: PMC3484208
- DOI: 10.1111/j.1600-6143.2012.04203.x
Intragraft CD11b(+) IDO(+) cells mediate cardiac allograft tolerance by ECDI-fixed donor splenocyte infusions
Abstract
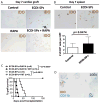
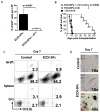
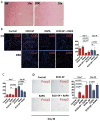
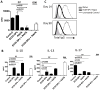
We have previously shown that pre- and post-transplant infusions of donor splenocytes treated with 1-ethyl-3-(3'-dimethylaminopropyl)-carbodiimide (ECDI-SPs) provide permanent donor-specific protection of islet allografts. The efficacy of donor ECDI-SPs in protecting vascularized cardiac allografts and mechanism(s) of protection are unknown. In this study, we show that infusions of ECDI-SPs significantly prolong cardiac allograft survival concomitant with an impressive accumulation of CD11b(+) IDO(+) cells in the cardiac allograft, and that the presence of this population is dependent on Gr1(+) cells. Consequently, depletion of Gr1(+) cells or inhibition of indoleamine 2,3 dioxygenase (IDO) activity abrogates graft protection by ECDI-SPs infusions. In addition, T cells from ECDI-SPs treated recipients secrete high levels of interleukin 10 and interleukin 13 upon in vitro restimulation, which are also dampened in recipients treated with the IDO inhibitor. Furthermore, combination of donor ECDI-SPs with a short course of rapamycin provides indefinite cardiac allograft survival in 100% of the recipients. These findings reveal a novel mechanism of donor ECDI-SPs in inducing cardiac transplant tolerance and provide several targets that are amenable to therapeutic manipulations for tolerance induction for cardiac transplantation.
© Copyright 2012 The American Society of Transplantation and the American Society of Transplant Surgeons.
Conflict of interest statement
The authors of this manuscript have no conflicts of interest to disclose as described by the
Figures

References
-
- Lechler RI, Sykes M, Thomson AW, Turka LA. Organ transplantation--how much of the promise has been realized? Nat Med. 2005 Jun;11(6):605–13. - PubMed
-
- Dugast AS, Haudebourg T, Coulon F, Heslan M, Haspot F, Poirier N, et al. Myeloid-derived suppressor cells accumulate in kidney allograft tolerance and specifically suppress effector T cell expansion. J Immunol. 2008 Jun 15;180(12):7898–906. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

