Tubby regulates microglial phagocytosis through MerTK
- PMID: 22884297
- PMCID: PMC3466361
- DOI: 10.1016/j.jneuroim.2012.07.009
Tubby regulates microglial phagocytosis through MerTK
Abstract
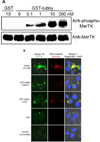
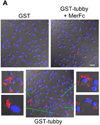
Immunologically-silent microglial phagocytosis of apoptotic cells and cellular debris is critical for CNS homeostasis and innate immune balance. The beneficial and detrimental effects of microglial phagocytosis on neurons remain controversial. Phagocytosis ligands are the key to selecting extracellular cargos, initiating the engulfment process, defining phagocyte functional roles and regulating phagocyte activities with therapeutic potentials. Here we characterized tubby as a new ligand to regulate microglial phagocytosis through MerTK receptor, which is well known for its immunosuppressive signaling. Tubby at 0.1nM significantly induced microglial phagocytosis of apoptotic cells with a maximal activity at 10nM. Tubby activated MerTK with receptor autophosphorylation in a similar dose range. Excessive soluble MerTK extracellular domain blocked tubby-mediated microglial phagocytosis of plasma membrane vesicles as cellular debris. Immunocytochemistry revealed that the ingested cargos were co-localized with MerTK-dependent non-muscle myosin II, whose rearrangement is necessary for cargo engulfment. Phagosome biomarker Rab7 was colocalized with cargos, suggesting that internalized cargos were targeted to phagocytic pathway. Tubby stimulated phagocytosis by neonatal and aged microglia with similar activities, but not by MerTK(-/-) microglia. These results suggest that tubby is a ligand to facilitate microglial phagocytosis through MerTK for the maintenance of CNS homeostasis.
Copyright © 2012 Elsevier B.V. All rights reserved.
Figures









Similar articles
-
Tubby and tubby-like protein 1 are new MerTK ligands for phagocytosis.EMBO J. 2010 Dec 1;29(23):3898-910. doi: 10.1038/emboj.2010.265. Epub 2010 Oct 26. EMBO J. 2010. PMID: 20978472 Free PMC article.
-
Galectin-3 is a new MerTK-specific eat-me signal.J Cell Physiol. 2012 Feb;227(2):401-7. doi: 10.1002/jcp.22955. J Cell Physiol. 2012. PMID: 21792939 Free PMC article.
-
MerTK Cleavage on Resident Cardiac Macrophages Compromises Repair After Myocardial Ischemia Reperfusion Injury.Circ Res. 2017 Sep 29;121(8):930-940. doi: 10.1161/CIRCRESAHA.117.311327. Epub 2017 Aug 29. Circ Res. 2017. PMID: 28851810 Free PMC article.
-
[Microglial Phagocytosis in the Neurodegenerative Diseases].Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2016 Apr;38(2):228-33. doi: 10.3881/j.issn.1000-503X.2016.02.018. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2016. PMID: 27181903 Review. Chinese.
-
Phagocytosis of microglia in the central nervous system diseases.Mol Neurobiol. 2014 Jun;49(3):1422-34. doi: 10.1007/s12035-013-8620-6. Epub 2014 Jan 7. Mol Neurobiol. 2014. PMID: 24395130 Free PMC article. Review.
Cited by
-
Glial responses during epileptogenesis in Mus musculus point to potential therapeutic targets.PLoS One. 2018 Aug 16;13(8):e0201742. doi: 10.1371/journal.pone.0201742. eCollection 2018. PLoS One. 2018. PMID: 30114263 Free PMC article.
-
Phagocyte dysfunction, tissue aging and degeneration.Ageing Res Rev. 2013 Sep;12(4):1005-12. doi: 10.1016/j.arr.2013.05.006. Epub 2013 Jun 4. Ageing Res Rev. 2013. PMID: 23748186 Free PMC article. Review.
-
YOD1 regulates microglial homeostasis by deubiquitinating MYH9 to promote the pathogenesis of Alzheimer's disease.Acta Pharm Sin B. 2025 Jan;15(1):331-348. doi: 10.1016/j.apsb.2024.11.020. Epub 2024 Nov 26. Acta Pharm Sin B. 2025. PMID: 40041897 Free PMC article.
-
The Phagocytic Code Regulating Phagocytosis of Mammalian Cells.Front Immunol. 2021 Jun 9;12:629979. doi: 10.3389/fimmu.2021.629979. eCollection 2021. Front Immunol. 2021. PMID: 34177884 Free PMC article. Review.
-
Mesd extrinsically promotes phagocytosis by retinal pigment epithelial cells.Cell Biol Toxicol. 2016 Aug;32(4):347-58. doi: 10.1007/s10565-016-9339-8. Epub 2016 May 17. Cell Biol Toxicol. 2016. PMID: 27184668 Free PMC article.
References
-
- Almolda B, Gonzalez B, Castellano B. Antigen presentation in EAE: role of microglia, macrophages and dendritic cells. Front Biosci. 2011;16:1157–1171. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

