Enhancement of dorsal hippocampal activity by knockdown of HCN1 channels leads to anxiolytic- and antidepressant-like behaviors
- PMID: 22884333
- PMCID: PMC3418514
- DOI: 10.1016/j.neuron.2012.05.027
Enhancement of dorsal hippocampal activity by knockdown of HCN1 channels leads to anxiolytic- and antidepressant-like behaviors
Abstract
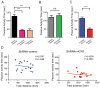
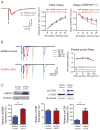
The hippocampus is an integral brain region for affective disorders. TRIP8b knockout mice lacking functional HCN channels as well as both HCN1 and HCN2 knockout mice have been shown to display antidepressant-like behaviors. The mechanisms or brain regions involved in these alterations in behavior, however, are not clear. We developed a lentiviral shRNA system to examine whether knockdown of HCN1 protein in the dorsal hippocampal CA1 region is sufficient to produce antidepressant-like effects. We found that knockdown of HCN1 channels increased cellular excitability and resulted in physiological changes consistent with a reduction of I(h). Rats infused with lentiviral shRNA-HCN1 in the dorsal hippocampal CA1 region displayed antidepressant- and anxiolytic-like behaviors associated with widespread enhancement of hippocampal activity and upregulation of BDNF-mTOR signaling pathways. Our results suggest that HCN1 protein could be a potential target for treatment of anxiety and depression disorders.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures






References
-
- Airan RD, Meltzer LA, Roy M, Gong Y, Chen H, Deisseroth K. High-speed imaging reveals neurophysiological links to behavior in an animal model of depression. Science. 2007;317:819–823. - PubMed
-
- Alldredge BK. Seizure risk associated with psychotropic drugs: clinical and pharmacokinetic considerations. Neurology. 1999;53:S 68–75. - PubMed
-
- Ball SG, Kuhn A, Wall D, Shekhar A, Goddard AW. Selective serotonin reuptake inhibitor treatment for generalized anxiety disorder: a double-blind, prospective comparison between paroxetine and sertraline. J Clin Psychiatry. 2005;66:94–99. - PubMed
-
- Berman RM, Cappiello A, Anand A, Oren DA, Heninger GR, Charney DS, Krystal JH. Antidepressant effects of ketamine in depressed patients. Biol Psychiatry. 2000;47:351–354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

