The accessory Sec protein Asp2 modulates GlcNAc deposition onto the serine-rich repeat glycoprotein GspB
- PMID: 22885294
- PMCID: PMC3458669
- DOI: 10.1128/JB.01000-12
The accessory Sec protein Asp2 modulates GlcNAc deposition onto the serine-rich repeat glycoprotein GspB
Abstract
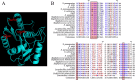
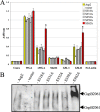
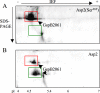
The accessory Sec system is a specialized transport system that exports serine-rich repeat (SRR) glycoproteins of Gram-positive bacteria. This system contains two homologues of the general secretory (Sec) pathway (SecA2 and SecY2) and several other essential proteins (Asp1 to Asp5) that share no homology to proteins of known function. In Streptococcus gordonii, Asp2 is required for the transport of the SRR adhesin GspB, but its role in export is unknown. Tertiary structure predictions suggest that the carboxyl terminus of Asp2 resembles the catalytic region of numerous enzymes that function through a Ser-Asp-His catalytic triad. Sequence alignment of all Asp2 homologues identified a highly conserved pentapeptide motif (Gly-X-Ser(362)-X-Gly) typical of most Ser-Asp-His catalytic triads, where Ser forms the reactive residue. Site-directed mutagenesis of residues comprising the predicted catalytic triad of Asp2 of S. gordonii had no effect upon GspB transport but did result in a marked change in the electrophoretic mobility of the protein. Lectin-binding studies and monosaccharide content analysis of this altered glycoform revealed an increase in glucosamine deposition. Random mutagenesis of the Asp2 region containing this catalytic domain also disrupted GspB transport. Collectively, our findings suggest that Asp2 is a bifunctional protein that is essential for both GspB transport and correct glycosylation. The catalytic domain may be responsible for controlling the glycosylation of GspB, while other surrounding regions are functionally required for glycoprotein transport.
Figures




Similar articles
-
Asp2 and Asp3 interact directly with GspB, the export substrate of the Streptococcus gordonii accessory Sec System.J Bacteriol. 2011 Jul;193(13):3165-74. doi: 10.1128/JB.00057-11. Epub 2011 Apr 29. J Bacteriol. 2011. PMID: 21531800 Free PMC article.
-
O-acetylation of the serine-rich repeat glycoprotein GspB is coordinated with accessory Sec transport.PLoS Pathog. 2017 Aug 21;13(8):e1006558. doi: 10.1371/journal.ppat.1006558. eCollection 2017 Aug. PLoS Pathog. 2017. PMID: 28827841 Free PMC article.
-
Membrane trafficking of the bacterial adhesin GspB and the accessory Sec transport machinery.J Biol Chem. 2019 Feb 1;294(5):1502-1515. doi: 10.1074/jbc.RA118.005657. Epub 2018 Dec 4. J Biol Chem. 2019. PMID: 30514759 Free PMC article.
-
The Two Distinct Types of SecA2-Dependent Export Systems.Microbiol Spectr. 2019 May;7(3):10.1128/microbiolspec.psib-0025-2018. doi: 10.1128/microbiolspec.PSIB-0025-2018. Microbiol Spectr. 2019. PMID: 31215505 Free PMC article. Review.
-
Glycosylation and biogenesis of a family of serine-rich bacterial adhesins.Microbiology (Reading). 2009 Feb;155(Pt 2):317-327. doi: 10.1099/mic.0.025221-0. Microbiology (Reading). 2009. PMID: 19202081 Review.
Cited by
-
The Plasmin-Sensitive Protein Pls in Methicillin-Resistant Staphylococcus aureus (MRSA) Is a Glycoprotein.PLoS Pathog. 2017 Jan 12;13(1):e1006110. doi: 10.1371/journal.ppat.1006110. eCollection 2017 Jan. PLoS Pathog. 2017. PMID: 28081265 Free PMC article.
-
Transcriptional organization of pneumococcal psrP-secY2A2 and impact of GtfA and GtfB deletion on PsrP-associated virulence properties.Microbes Infect. 2017 Jun;19(6):323-333. doi: 10.1016/j.micinf.2017.04.001. Epub 2017 Apr 10. Microbes Infect. 2017. PMID: 28408270 Free PMC article.
-
Defining the enzymatic pathway for polymorphic O-glycosylation of the pneumococcal serine-rich repeat protein PsrP.J Biol Chem. 2017 Apr 14;292(15):6213-6224. doi: 10.1074/jbc.M116.770446. Epub 2017 Feb 28. J Biol Chem. 2017. PMID: 28246170 Free PMC article.
-
Gap2 promotes the formation of a stable protein complex required for mature Fap1 biogenesis.J Bacteriol. 2013 May;195(10):2166-76. doi: 10.1128/JB.02255-12. Epub 2013 Mar 8. J Bacteriol. 2013. PMID: 23475979 Free PMC article.
-
Serine-rich repeat proteins: well-known yet little-understood bacterial adhesins.J Bacteriol. 2024 Jan 25;206(1):e0024123. doi: 10.1128/jb.00241-23. Epub 2023 Nov 17. J Bacteriol. 2024. PMID: 37975670 Free PMC article. Review.
References
-
- Amada K, Haruki M, Imanaka T, Morikawa M, Kanaya S. 2000. Overproduction in Escherichia coli, purification and characterization of a family I.3 lipase from Pseudomonas sp. MIS38. Biochim. Biophys. Acta 1478:201–210 - PubMed
-
- Bensing BA, Sullam PM. 2002. An accessory sec locus of Streptococcus gordonii is required for export of the surface protein GspB and for normal levels of binding to human platelets. Mol. Microbiol. 44:1081–1094 - PubMed

