Targeted oxidation of Torpedo californica acetylcholinesterase by singlet oxygen: identification of N-formylkynurenine tryptophan derivatives within the active-site gorge of its complex with the photosensitizer methylene blue
- PMID: 22888904
- PMCID: PMC3613852
- DOI: 10.1042/BJ20120992
Targeted oxidation of Torpedo californica acetylcholinesterase by singlet oxygen: identification of N-formylkynurenine tryptophan derivatives within the active-site gorge of its complex with the photosensitizer methylene blue
Abstract
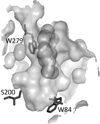
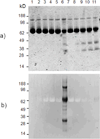
The principal role of AChE (acetylcholinesterase) is termination of impulse transmission at cholinergic synapses by rapid hydrolysis of the neurotransmitter acetylcholine. The active site of AChE is near the bottom of a long and narrow gorge lined with aromatic residues. It contains a CAS (catalytic 'anionic' subsite) and a second PAS (peripheral 'anionic' site), the gorge mouth, both of which bind acetylcholine via π-cation interactions, primarily with two conserved tryptophan residues. It was shown previously that generation of (1)O(2) by illumination of MB (Methylene Blue) causes irreversible inactivation of TcAChE (Torpedo californica AChE), and suggested that photo-oxidation of tryptophan residues might be responsible. In the present study, structural modification of the TcAChE tryptophan residues induced by MB-sensitized oxidation was investigated using anti-N-formylkynurenine antibodies and MS. From these analyses, we determined that N-formylkynurenine derivatives were specifically produced from Trp(84) and Trp(279), present at the CAS and PAS respectively. Peptides containing these two oxidized tryptophan residues were not detected when the competitive inhibitors, edrophonium and propidium (which should displace MB from the gorge) were present during illumination, in agreement with their efficient protection against the MB-induced photo-inactivation. Thus the bound MB elicited selective action of (1)O(2) on the tryptophan residues facing on to the water-filled active-site gorge. The findings of the present study thus demonstrate the localized action and high specificity of MB-sensitized photo-oxidation of TcAChE, as well as the value of this enzyme as a model system for studying the mechanism of action and specificity of photosensitizing agents.
Figures



Similar articles
-
Structural and functional characterization of the interaction of the photosensitizing probe methylene blue with Torpedo californica acetylcholinesterase.Protein Sci. 2012 Aug;21(8):1138-52. doi: 10.1002/pro.2101. Epub 2012 Jun 26. Protein Sci. 2012. PMID: 22674800 Free PMC article.
-
The specific interaction of the photosensitizer methylene blue with acetylcholinesterase provides a model system for studying the molecular consequences of photodynamic therapy.Chem Biol Interact. 2013 Mar 25;203(1):63-6. doi: 10.1016/j.cbi.2012.10.021. Epub 2012 Nov 15. Chem Biol Interact. 2013. PMID: 23159732 Free PMC article.
-
Targeted oxidation of Torpedo californica acetylcholinesterase by singlet oxygen.Photochem Photobiol. 2011 Mar-Apr;87(2):308-16. doi: 10.1111/j.1751-1097.2010.00857.x. Epub 2010 Dec 13. Photochem Photobiol. 2011. PMID: 21155827
-
A preliminary comparison of structural models for catalytic intermediates of acetylcholinesterase.Chem Biol Interact. 1999 May 14;119-120:43-52. doi: 10.1016/s0009-2797(99)00012-5. Chem Biol Interact. 1999. PMID: 10421437 Review.
-
[The fasciculin-acetylcholinesterase interaction].J Soc Biol. 1999;193(6):505-8. J Soc Biol. 1999. PMID: 10783708 Review. French.
Cited by
-
Hypericin-mediated photooxidative damage of α-crystallin in human lens epithelial cells.Free Radic Biol Med. 2013 Jul;60:347-54. doi: 10.1016/j.freeradbiomed.2013.02.023. Epub 2013 Feb 27. Free Radic Biol Med. 2013. PMID: 23453985 Free PMC article.
-
Structural and functional characterization of the interaction of the photosensitizing probe methylene blue with Torpedo californica acetylcholinesterase.Protein Sci. 2012 Aug;21(8):1138-52. doi: 10.1002/pro.2101. Epub 2012 Jun 26. Protein Sci. 2012. PMID: 22674800 Free PMC article.
-
The specific interaction of the photosensitizer methylene blue with acetylcholinesterase provides a model system for studying the molecular consequences of photodynamic therapy.Chem Biol Interact. 2013 Mar 25;203(1):63-6. doi: 10.1016/j.cbi.2012.10.021. Epub 2012 Nov 15. Chem Biol Interact. 2013. PMID: 23159732 Free PMC article.
-
Tripping up Trp: Modification of protein tryptophan residues by reactive oxygen species, modes of detection, and biological consequences.Free Radic Biol Med. 2015 Dec;89:220-8. doi: 10.1016/j.freeradbiomed.2015.08.003. Epub 2015 Sep 21. Free Radic Biol Med. 2015. PMID: 26393422 Free PMC article. Review.
-
The impact of crystallization conditions on structure-based drug design: A case study on the methylene blue/acetylcholinesterase complex.Protein Sci. 2016 Jun;25(6):1096-114. doi: 10.1002/pro.2923. Epub 2016 Mar 28. Protein Sci. 2016. PMID: 26990888 Free PMC article.
References
-
- Silman I, Sussman JL. Acetylcholinesterase: ‘Classical’ and ‘non-classical’ functions and pharmacology. Curr. Opin. Pharmacol. 2005;5:293–302. - PubMed
-
- Hasinoff BB. Kinetics of acetylthiocholine binding to electric eel acetylcholinesterase in glycerol/water solvents of increased viscosity. Evidence for a diffusion-controlled reaction. Biochim. Biophys. Acta. 1982;704:52–58. - PubMed
-
- Koelle GB. Cholinesterases and anticholinesterase agents. Berlin: Springer-Verlag; 1963. Cytological distributions and physiological functions of cholinesterases; pp. 187–298.
-
- Azevedo Marques L, Giera M, Lingeman H, Niessen WMA. Analysis of acetylcholinesterase inhibitors: bioanalysis, degradation and metabolism. Biomed. Chromat. 2011;25:278–299. - PubMed
-
- Reinhard S, Thomas A. The cholinergic system in aging and neuronal degeneration. Behavioural in brain research. 2011;221:555–563. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

