Microbial carriage state of peripheral blood dendritic cells (DCs) in chronic periodontitis influences DC differentiation, atherogenic potential
- PMID: 22891282
- PMCID: PMC3459682
- DOI: 10.4049/jimmunol.1201053
Microbial carriage state of peripheral blood dendritic cells (DCs) in chronic periodontitis influences DC differentiation, atherogenic potential
Abstract
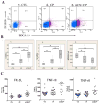
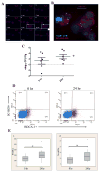
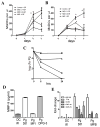
The low-grade oral infection chronic periodontitis (CP) has been implicated in coronary artery disease risk, but the mechanisms are unclear. In this study, a pathophysiological role for blood dendritic cells (DCs) in systemic dissemination of oral mucosal pathogens to atherosclerotic plaques was investigated in humans. The frequency and microbiome of CD19(-)BDCA-1(+)DC-SIGN(+) blood myeloid DCs (mDCs) were analyzed in CP subjects with or without existing acute coronary syndrome and in healthy controls. FACS analysis revealed a significant increase in blood mDCs in the following order: healthy controls < CP < acute coronary syndrome/CP. Analysis of the blood mDC microbiome by 16S rDNA sequencing showed Porphyromonas gingivalis and other species, including (cultivable) Burkholderia cepacia. The mDC carriage rate with P. gingivalis correlated with oral carriage rate and with serologic exposure to P. gingivalis in CP subjects. Intervention (local debridement) to elicit a bacteremia increased the mDC carriage rate and frequency in vivo. In vitro studies established that P. gingivalis enhanced by 28% the differentiation of monocytes into immature mDCs; moreover, mDCs secreted high levels of matrix metalloproteinase-9 and upregulated C1q, heat shock protein 60, heat shock protein 70, CCR2, and CXCL16 transcripts in response to P. gingivalis in a fimbriae-dependent manner. Moreover, the survival of the anaerobe P. gingivalis under aerobic conditions was enhanced when within mDCs. Immunofluorescence analysis of oral mucosa and atherosclerotic plaques demonstrate infiltration with mDCs, colocalized with P. gingivalis. Our results suggest a role for blood mDCs in harboring and disseminating pathogens from oral mucosa to atherosclerosis plaques, which may provide key signals for mDC differentiation and atherogenic conversion.
Conflict of interest statement
The authors have no financial conflicts of interest
Figures


References
-
- Naghavi M, Falk E, Hecht HS, Jamieson MJ, Kaul S, Berman D, Fayad Z, Budoff MJ, Rumberger J, Naqvi TZ, Shaw LJ, Faergeman O, Cohn J, Bahr R, Koenig W, Demirovic J, Arking D, Herrera VL, Badimon J, Goldstein JA, Rudy Y, Airaksinen J, Schwartz RS, Riley WA, Mendes RA, Douglas P, Shah PK. From vulnerable plaque to vulnerable patient--Part III: Executive summary of the Screening for Heart Attack Prevention and Education (SHAPE) Task Force report. Am J Cardiol. 2006;98:2H–15H. - PubMed
-
- Vita JA, Loscalzo J. Shouldering the risk factor burden: infection, atherosclerosis, and the vascular endothelium. Circulation. 2002;106:164–166. - PubMed
-
- Pesonen E, El-Segaier M, Persson K, Puolakkainen M, Sarna S, Ohlin H, Pussinen PJ. Infections as a stimulus for coronary occlusion, obstruction, or acute coronary syndromes. Ther Adv Cardiovasc Dis. 2009;3:447–454. - PubMed
-
- Gibson FC, 3rd, Yumoto H, Takahashi Y, Chou HH, Genco CA. Innate immune signaling and Porphyromonas gingivalis-accelerated atherosclerosis. J Dent Res. 2006;85:106–121. - PubMed
-
- Offenbacher S, Beck JD, Moss K, Mendoza L, Paquette DW, Barrow DA, Couper DJ, Stewart DD, Falkner KL, Graham SP, Grossi S, Gunsolley JC, Madden T, Maupome G, Trevisan M, Van Dyke TE, Genco RJ. Results from the Periodontitis and Vascular Events (PAVE) Study: a pilot multicentered, randomized, controlled trial to study effects of periodontal therapy in a secondary prevention model of cardiovascular disease. J Periodontol. 2009;80:190–201. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

