Prediction of free imatinib concentrations based on total plasma concentrations in patients with gastrointestinal stromal tumours
- PMID: 22891806
- PMCID: PMC3612719
- DOI: 10.1111/j.1365-2125.2012.04422.x
Prediction of free imatinib concentrations based on total plasma concentrations in patients with gastrointestinal stromal tumours
Abstract
Aim: Total imatinib concentrations are currently measured for the therapeutic drug monitoring of imatinib, whereas only free drug equilibrates with cells for pharmacological action. Due to technical and cost limitations, routine measurement of free concentrations is generally not performed. In this study, free and total imatinib concentrations were measured to establish a model allowing the confident prediction of imatinib free concentrations based on total concentrations and plasma proteins measurements.
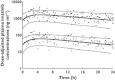
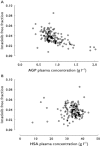
Methods: One hundred and fifty total and free plasma concentrations of imatinib were measured in 49 patients with gastrointestinal stromal tumours. A population pharmacokinetic model was built up to characterize mean total and free concentrations with inter-patient and intrapatient variability, while taking into account α1 -acid glycoprotein (AGP) and human serum albumin (HSA) concentrations, in addition to other demographic and environmental covariates.
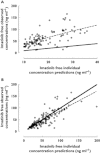
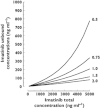
Results: A one compartment model with first order absorption was used to characterize total and free imatinib concentrations. Only AGP influenced imatinib total clearance. Imatinib free concentrations were best predicted using a non-linear binding model to AGP, with a dissociation constant Kd of 319 ng ml(-1) , assuming a 1:1 molar binding ratio. The addition of HSA in the equation did not improve the prediction of imatinib unbound concentrations.
Conclusion: Although free concentration monitoring is probably more appropriate than total concentrations, it requires an additional ultrafiltration step and sensitive analytical technology, not always available in clinical laboratories. The model proposed might represent a convenient approach to estimate imatinib free concentrations. However, therapeutic ranges for free imatinib concentrations remain to be established before it enters into routine practice.
© 2012 The Authors. British Journal of Clinical Pharmacology © 2012 The British Pharmacological Society.
Figures

Similar articles
-
Therapeutic drug monitoring of imatinib: Bayesian and alternative methods to predict trough levels.Clin Pharmacokinet. 2012 Mar 1;51(3):187-201. doi: 10.2165/11596990-000000000-00000. Clin Pharmacokinet. 2012. PMID: 22339450
-
Association of ABCG2 polymorphism with clinical efficacy of imatinib in patients with gastrointestinal stromal tumor.Cancer Chemother Pharmacol. 2015 Jan;75(1):173-82. doi: 10.1007/s00280-014-2630-6. Epub 2014 Nov 23. Cancer Chemother Pharmacol. 2015. PMID: 25417047
-
[Potential clinical benefit of therapeutic drug monitoring of imatinib in oncology].Klin Onkol. 2015;28(2):105-11. doi: 10.14735/amko2015105. Klin Onkol. 2015. PMID: 25882020 Review. Czech.
-
Imatinib plasma levels during successful long-term treatment of metastatic-gastrointestinal stromal tumors.Hepatogastroenterology. 2014 Oct;61(135):1984-9. Hepatogastroenterology. 2014. PMID: 25713899
-
Imatinib mesylate in the treatment of gastrointestinal stromal tumour.Expert Opin Pharmacother. 2005 Jan;6(1):105-13. doi: 10.1517/14656566.6.1.105. Expert Opin Pharmacother. 2005. PMID: 15709888 Review.
Cited by
-
Precision Medicine in Oncology: Imatinib Dosing in the Obese Cancer Population Using Virtual Clinical Trials.CPT Pharmacometrics Syst Pharmacol. 2025 Jun;14(6):1050-1064. doi: 10.1002/psp4.70018. Epub 2025 Mar 27. CPT Pharmacometrics Syst Pharmacol. 2025. PMID: 40150877 Free PMC article.
-
Practical guidelines for therapeutic drug monitoring of anticancer tyrosine kinase inhibitors: focus on the pharmacokinetic targets.Clin Pharmacokinet. 2014 Apr;53(4):305-25. doi: 10.1007/s40262-014-0137-2. Clin Pharmacokinet. 2014. PMID: 24566736 Review.
-
Towards point of care systems for the therapeutic drug monitoring of imatinib.Anal Bioanal Chem. 2020 Sep;412(24):5925-5933. doi: 10.1007/s00216-020-02545-4. Epub 2020 Mar 12. Anal Bioanal Chem. 2020. PMID: 32166445 Review.
-
Physiologically Based Pharmacokinetic Modelling to Predict Imatinib Exposures in Cancer Patients with Renal Dysfunction: A Case Study.Pharmaceutics. 2023 Jul 11;15(7):1922. doi: 10.3390/pharmaceutics15071922. Pharmaceutics. 2023. PMID: 37514108 Free PMC article.
-
The Steps to Therapeutic Drug Monitoring: A Structured Approach Illustrated With Imatinib.Front Pharmacol. 2020 Mar 3;11:177. doi: 10.3389/fphar.2020.00177. eCollection 2020. Front Pharmacol. 2020. PMID: 32194413 Free PMC article. Review.
References
-
- Ceciliani F, Pocacqua V. The acute phase protein alpha1-acid glycoprotein: a model for altered glycosylation during diseases. Curr Protein Pept Sci. 2007;8:91–108. - PubMed
-
- Fitos I, Visy J, Zsila F, Mady G, Simonyi M. Selective binding of imatinib to the genetic variants of human alpha1-acid glycoprotein. Biochim Biophys Acta. 2006;1760:1704–1712. - PubMed
-
- Gambacorti-Passerini C, Barni R, le Cortre CP, Zucchetti M, Cabrita G, Cleris L, Rossi F, Gianazza E, Brueggen J, Cozens R, Pioltelli P, Pogliani E, Corneo G, Formelli F, D'Incalci M. Role of alpha1 acid glycoprotein in the in vivo resistance of human BCR-ABL(+) leukemic cells to the abl inhibitor STI571. J Natl Cancer Inst. 2000;92:1641–1650. - PubMed
-
- Kretz O, Weiss HM, Schumacher MM, Gross G. In vitro blood distribution and plasma protein binding of the tyrosine kinase inhibitor imatinib and its active metabolite, CGP74588, in rat, mouse, dog, monkey, healthy humans and patients with acute lymphatic leukaemia. Br J Clin Pharmacol. 2004;58:212–216. - PMC - PubMed
-
- Smith KD, Paterson S. Binding of alpha-1-acid glycoprotein to imatinib following increased dosage of drug. Haematologica. 2005;90(Suppl):ELT01. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

