Targeting TCTP as a new therapeutic strategy in castration-resistant prostate cancer
- PMID: 22893039
- PMCID: PMC3519986
- DOI: 10.1038/mt.2012.155
Targeting TCTP as a new therapeutic strategy in castration-resistant prostate cancer
Abstract
Heat shock protein 27 (Hsp27) is highly overexpressed in castration-resistant prostate cancer (CRPC) and an antisense inhibitor (OGX-427) is currently in phase II clinical trials. In order to understand mechanisms of action of Hsp27 and find new therapeutic targets specific of CRPC, we screened for Hsp27 client proteins. Here, we report that translationally controlled tumor protein (TCTP) is a new Hsp27 client protein involved in Hsp27 cytoprotection. We found that TCTP expression is absent or weak in normal prostate cells, moderately expressed in 18.5% of treatment naive PC, and becomes uniformly and strongly expressed in 75% of CRPC. To define TCTP function, we developed and worldwide patented a TCTP antisense oligonucleotide (ASO). Interestingly, we found that CRPC progression correlates with TCTP overexpression and loss of P53. TCTP knockdown restored P53 expression and function, suggesting that castration-sensitivity is directly linked to P53 expression. Collectively, these findings provide a new Hsp27 cytoprotection mechanism in CRPC, and preclinical proof-of-concept that combining ASO-mediated TCTP knockdown with castration and/or docetaxel therapy could serve as a novel strategy to treat CRPC, with no or little toxicity for normal prostate cells.
Figures
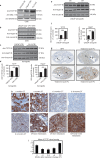

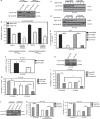

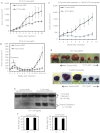
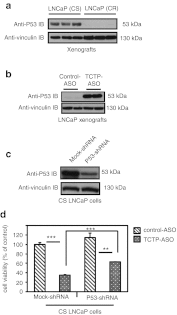
References
-
- Calderwood SK, Khaleque MA, Sawyer DB., and, Ciocca DR. Heat shock proteins in cancer: chaperones of tumorigenesis. Trends Biochem Sci. 2006;31:164–172. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

