Galactose metabolism plays a crucial role in biofilm formation by Bacillus subtilis
- PMID: 22893383
- PMCID: PMC3419520
- DOI: 10.1128/mBio.00184-12
Galactose metabolism plays a crucial role in biofilm formation by Bacillus subtilis
Erratum in
- MBio. 2013;4(1). doi:10.1128/mBio.00555-12
Abstract
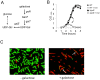
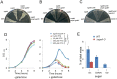
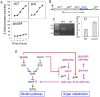
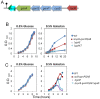
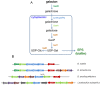
Galactose is a common monosaccharide that can be utilized by all living organisms via the activities of three main enzymes that make up the Leloir pathway: GalK, GalT, and GalE. In Bacillus subtilis, the absence of GalE causes sensitivity to exogenous galactose, leading to rapid cell lysis. This effect can be attributed to the accumulation of toxic galactose metabolites, since the galE mutant is blocked in the final step of galactose catabolism. In a screen for suppressor mutants restoring viability to a galE null mutant in the presence of galactose, we identified mutations in sinR, which is the major biofilm repressor gene. These mutations caused an increase in the production of the exopolysaccharide (EPS) component of the biofilm matrix. We propose that UDP-galactose is the toxic galactose metabolite and that it is used in the synthesis of EPS. Thus, EPS production can function as a shunt mechanism for this toxic molecule. Additionally, we demonstrated that galactose metabolism genes play an essential role in B. subtilis biofilm formation and that the expressions of both the gal and eps genes are interrelated. Finally, we propose that B. subtilis and other members of the Bacillus genus may have evolved to utilize naturally occurring polymers of galactose, such as galactan, as carbon sources.
Importance: Bacteria switch from unicellular to multicellular states by producing extracellular matrices that contain exopolysaccharides. In such aggregates, known as biofilms, bacteria are more resistant to antibiotics. This makes biofilms a serious problem in clinical settings. The resilience of biofilms makes them very useful in industrial settings. Thus, understanding the production of biofilm matrices is an important problem in microbiology. In studying the synthesis of the biofilm matrix of Bacillus subtilis, we provide further understanding of a long-standing microbiological observation that certain mutants defective in the utilization of galactose became sensitive to it. In this work, we show that the toxicity observed before was because cells were grown under conditions that were not propitious to produce the exopolysaccharide component of the matrix. When cells are grown under conditions that favor matrix production, the toxicity of galactose is relieved. This allowed us to demonstrate that galactose metabolism is essential for the synthesis of the extracellular matrix.
Figures

References
-
- Branda SS, Vik A, Friedman L, Kolter R. 2005. Biofilms: the matrix revisited. Trends Microbiol. 13:20–26 - PubMed
-
- Branda SS, Chu F, Kearns DB, Losick R, Kolter R. 2006. A major protein component of the Bacillus subtilis biofilm matrix. Mol. Microbiol. 59:1229–1238 - PubMed
-
- Kearns DB, Chu F, Branda SS, Kolter R, Losick R. 2005. A master regulator for biofilm formation by Bacillus subtilis. Mol. Microbiol. 55:739–749 - PubMed
-
- Flemming HC, Wingender J. 2010. The biofilm matrix. Nat. Rev. Microbiol. 8:623–633 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
