Macrophages promote fibroblast growth factor receptor-driven tumor cell migration and invasion in a CXCR2-dependent manner
- PMID: 22893608
- PMCID: PMC3553584
- DOI: 10.1158/1541-7786.MCR-12-0275
Macrophages promote fibroblast growth factor receptor-driven tumor cell migration and invasion in a CXCR2-dependent manner
Abstract
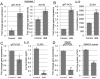
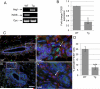
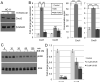
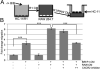
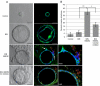
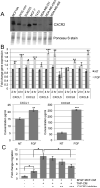
Infiltration of immune cells, specifically macrophages, into the tumor microenvironment has been linked to increased mammary tumor formation and progression. Activation of growth factor receptor signaling pathways within mammary epithelial cells, such as the fibroblast growth factor receptor 1 (FGFR1) pathway, induces recruitment of macrophages to the mammary epithelium. These macrophages promote increased epithelial cell proliferation and angiogenesis. However, the specific mechanisms by which these macrophages are regulated by the preneoplastic epithelial cells and the mechanisms of action of the macrophages within the developing FGFR1-driven tumor microenvironment remain unknown. In this study, we show that activation of inducible FGFR1 in mammary glands leads to decreased activity of the TGFβ/Smad3 pathway in macrophages associated with early stage lesions. Further studies show that macrophages have increased expression of inflammatory chemokines that bind Cxcr2 following exposure to conditioned media from mammary epithelial and tumor cells in which the FGF pathway had been activated. The increase in these ligands is inhibited following activation of the TGFβ pathway, suggesting that decreased TGFβ signaling contributes to the upregulation of these chemokines. Using coculture studies, we further show that macrophages are capable of promoting epithelial and tumor cell migration and invasion through activation of Cxcr2. These results indicate that macrophage-derived Cxcr2 ligands may be important for promoting mammary tumor formation regulated by FGFR signaling. Furthermore, these results suggest that targeting Cxcr2 may represent a novel therapeutic strategy for breast cancers that are associated with high levels of infiltrating macrophages.
Figures
References
-
- de Visser KE, Eichten A, Coussens LM. Paradoxical roles of the immune system during cancer development. Nat Rev Cancer. 2006;6(1):24–37. - PubMed
-
- Bingle L, Brown NJ, Lewis CE. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol. 2002;196(3):254–65. - PubMed
-
- Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer. 2004;4(1):71–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

