Cytoplasmic superoxide radical: a possible contributing factor to intracellular Aβ oligomerization in Alzheimer disease
- PMID: 22896786
- PMCID: PMC3419108
- DOI: 10.4161/cib.19548
Cytoplasmic superoxide radical: a possible contributing factor to intracellular Aβ oligomerization in Alzheimer disease
Abstract
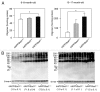
Soluble amyloid β (Aβ) oligomers cause memory loss and synaptic dysfunction in Alzheimer disease (AD). Despite intensive studies on Aβ assembly in vitro and in vivo, the localization and cellular mechanism of Aβ oligomerization are not fully understood. Previously, we demonstrated that cytoplasmic superoxide radicals contribute to drusen deposition, a hallmark of age-related macular degeneration as well as other geriatric diseases (fatty liver, skin thinning, and osteoporosis). Using a transgenic mouse model of AD, we recently clarified the role of cytoplasmic oxidative stress in cognitive impairment and oligomer formation. Moreover, we also found that these phenomena were associated with neuroinflammation, tau phosphorylation, and synaptic loss. Notably, studies using human brains support the involvement of cytoplasmic superoxide radicals in AD pathology. In this addendum to Murakami et al. (JBC 2011), we discuss and comment on intracellular Aβ oligomer formation and the possible therapeutic effects of intracellular redox state modulators.
Keywords: Alzheimer’s disease; SOD; amyloid β; memory loss; oligomer; oxidative stress; superoxide radical.
Figures
References
LinkOut - more resources
Full Text Sources
