New insights on stromules: stroma filled tubules extended by independent plastids
- PMID: 22899053
- PMCID: PMC3489645
- DOI: 10.4161/psb.21342
New insights on stromules: stroma filled tubules extended by independent plastids
Abstract
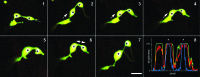
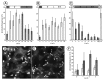
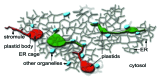
The recognition of stromules as sporadically extended stroma filled tubules from all kinds of plastids constitutes one of the major insights that resulted from the direct application of green fluorescent protein aided imaging of living plant cells. Observations of dynamic green fluorescent stromules strongly suggested that plastids frequently interact with each other while photo-bleaching of interconnected plastids indicated that proteins can move within the stroma filled tubules. These observations readily fit into the prevailing concept of the endosymbiogenic origins of plastids and provided stromules the status of conduits for inter-plastid communication and macromolecule transfer. However, experimental evidence obtained recently through the use of photoconvertible protein labeled stromules strongly supports plastid independence rather than their interconnectivity. Additional information on stress conditions inducing stromules and observations on their alignment with other organelles suggests that the major role of stromules is to increase the interactive surface of a plastid with the rest of the cytoplasm.
Figures

Similar articles
-
The myth of interconnected plastids and related phenomena.Protoplasma. 2015 Jan;252(1):359-71. doi: 10.1007/s00709-014-0666-4. Epub 2014 Jun 26. Protoplasma. 2015. PMID: 24965372
-
Exclusion of plastid nucleoids and ribosomes from stromules in tobacco and Arabidopsis.Plant J. 2012 Feb;69(3):399-410. doi: 10.1111/j.1365-313X.2011.04798.x. Epub 2011 Nov 8. Plant J. 2012. PMID: 21951134
-
In vivo analysis of interactions between GFP-labeled microfilaments and plastid stromules.BMC Plant Biol. 2004 Feb 10;4:2. doi: 10.1186/1471-2229-4-2. BMC Plant Biol. 2004. PMID: 15018639 Free PMC article.
-
Trafficking of proteins through plastid stromules.Plant Cell. 2013 Aug;25(8):2774-82. doi: 10.1105/tpc.113.112870. Epub 2013 Aug 27. Plant Cell. 2013. PMID: 23983219 Free PMC article. Review.
-
Stromules and the dynamic nature of plastid morphology.J Microsc. 2004 May;214(Pt 2):124-37. doi: 10.1111/j.0022-2720.2004.01317.x. J Microsc. 2004. PMID: 15102061 Review.
Cited by
-
Organelle extensions in plant cells.Plant Physiol. 2021 Apr 2;185(3):593-607. doi: 10.1093/plphys/kiaa055. Plant Physiol. 2021. PMID: 33793902 Free PMC article.
-
Fluorescent protein flow within stromules.Plant Cell. 2013 Aug;25(8):2771-2. doi: 10.1105/tpc.113.117416. Epub 2013 Aug 27. Plant Cell. 2013. PMID: 23983222 Free PMC article. No abstract available.
-
Chloroplasts extend stromules independently and in response to internal redox signals.Proc Natl Acad Sci U S A. 2015 Aug 11;112(32):10044-9. doi: 10.1073/pnas.1511570112. Epub 2015 Jul 6. Proc Natl Acad Sci U S A. 2015. PMID: 26150490 Free PMC article.
-
The myth of interconnected plastids and related phenomena.Protoplasma. 2015 Jan;252(1):359-71. doi: 10.1007/s00709-014-0666-4. Epub 2014 Jun 26. Protoplasma. 2015. PMID: 24965372
-
Fluorescent Protein Aided Insights on Plastids and their Extensions: A Critical Appraisal.Front Plant Sci. 2016 Jan 20;6:1253. doi: 10.3389/fpls.2015.01253. eCollection 2015. Front Plant Sci. 2016. PMID: 26834765 Free PMC article. Review.
References
-
- Haberlandt G. Die chlorophyllkorper der selaginellen. Flora. 1888;71:291–308.
-
- Menzel D. An interconnected plastidom in Acetabularia: Implications for the mechanism of chloroplast motility. Protoplasma. 1994;179:166–71. doi: 10.1007/BF01403955. - DOI
-
- Köhler RH, Hanson MR. Plastid tubules of higher plants are tissue-specific and developmentally regulated. J Cell Sci. 2000;113:81–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
