Mitochondria-mediated energy adaption in cancer: the H(+)-ATP synthase-geared switch of metabolism in human tumors
- PMID: 22901241
- PMCID: PMC3691914
- DOI: 10.1089/ars.2012.4883
Mitochondria-mediated energy adaption in cancer: the H(+)-ATP synthase-geared switch of metabolism in human tumors
Abstract
Significance: Since the signing of the National Cancer Act in 1971, cancer still remains a major cause of death despite significant progresses made in understanding the biology and treatment of the disease. After many years of ostracism, the peculiar energy metabolism of tumors has been recognized as an additional phenotypic trait of the cancer cell.
Recent advances: While the enhanced aerobic glycolysis of carcinomas has already been translated to bedside for precise tumor imaging and staging of cancer patients, accepting that an impaired bioenergetic function of mitochondria is pivotal to understand energy metabolism of tumors and in its progression is debated. However, mitochondrial bioenergetics and cell death are tightly connected.
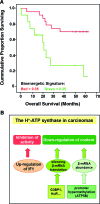
Critical issues: Recent clinical findings indicate that H(+)-ATP synthase, a core component of mitochondrial oxidative phosphorylation, is repressed at both the protein and activity levels in human carcinomas. This review summarizes the relevance that mitochondrial function has to understand energy metabolism of tumors and explores the connection between the bioenergetic function of the organelle and the activity of mitochondria as tumor suppressors.
Future directions: The reversible nature of energy metabolism in tumors highlights the relevance that the microenvironment has for tumor progression. Moreover, the stimulation of mitochondrial activity or the inhibition of glycolysis suppresses tumor growth. Future research should elucidate the mechanisms promoting the silencing of oxidative phosphorylation in carcinomas. The aim is the development of new therapeutic strategies tackling energy metabolism to eradicate tumors or at least, to maintain tumor dormancy and transform cancer into a chronic disease.
Figures

References
-
- Alavian KN. Li H. Collis L. Bonanni L. Zeng L. Sacchetti S. Lazrove E. Nabili P. Flaherty B. Graham M. Chen Y. Messerli SM. Mariggio MA. Rahner C. McNay E. Shore GC. Smith PJ. Hardwick JM. Jonas EA. Bcl-x(L) regulates metabolic efficiency of neurons through interaction with the mitochondrial F(1)F(O) ATP synthase. Nat Cell Biol. 2011;13:1224–1233. - PMC - PubMed
-
- Aldea M. Clofent J. Nunez de Arenas C. Chamorro M. Velasco M. Berrendero JR. Navarro C. Cuezva JM. Reverse phase protein microarrays quantify and validate the bioenergetic signature as biomarker in colorectal cancer. Cancer Lett. 2011;311:210–218. - PubMed
-
- Arismendi-Morillo G. Electron microscopy morphology of the mitochondrial network in gliomas and their vascular microenvironment. Biochim Biophys Acta. 2011;1807:602–608. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

