Exo- and endoribonucleolytic activities of yeast cytoplasmic and nuclear RNA exosomes are dependent on the noncatalytic core and central channel
- PMID: 22902556
- PMCID: PMC3472098
- DOI: 10.1016/j.molcel.2012.07.012
Exo- and endoribonucleolytic activities of yeast cytoplasmic and nuclear RNA exosomes are dependent on the noncatalytic core and central channel
Abstract
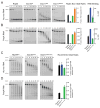
The RNA exosome is an essential multisubunit ribonuclease (RNase) that contributes to cytoplasmic and nuclear RNA decay and quality control. The 9-subunit exosome core (Exo9) features a prominent central channel formed by stacked asymmetric rings of six RNase PH-like proteins and three S1/KH domain proteins. Exo9 is catalytically inert but associates with Rrp44, an endoribonuclease and processive 3'→5' exoribonuclease, and Rrp6, a distributive 3'→5' exoribonuclease. We show that Exo9 and its central channel modulate all three yeast exosome RNase activities because channel occlusion attenuates RNA binding and RNase activities in vitro and fails to complement exosome functions in vivo. We find that Rrp6 stimulates Rrp44 RNase activities and that Rrp6 is inhibited by a mutation in the Rrp44 exoribonuclease active site in 11-subunit nuclear exosomes. These results suggest the exosome core and central channel is essential because it modulates each of the known RNase activities of the yeast RNA exosome.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures

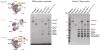




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

