The phytochrome-interacting vascular plant one-zinc finger1 and VOZ2 redundantly regulate flowering in Arabidopsis
- PMID: 22904146
- PMCID: PMC3462629
- DOI: 10.1105/tpc.112.101915
The phytochrome-interacting vascular plant one-zinc finger1 and VOZ2 redundantly regulate flowering in Arabidopsis
Abstract
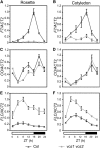
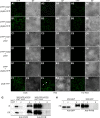
The timing of the transition to flowering in plants is regulated by various environmental factors, including daylength and light quality. Although the red/far-red photoreceptor phytochrome B (phyB) represses flowering by indirectly regulating the expression of a key flowering regulator, FLOWERING LOCUS T (FT), the mechanism of phyB signaling for flowering is largely unknown. Here, we identified two Arabidopsis thaliana genes, VASCULAR PLANT ONE-ZINC FINGER1 (VOZ1) and VOZ2, which are highly conserved throughout land plant evolution, as phyB-interacting factors. voz1 voz2 double mutants, but neither single mutant, showed a late-flowering phenotype under long-day conditions, which indicated that VOZ1 and VOZ2 redundantly promote flowering. voz1 voz2 mutations suppressed the early-flowering phenotype of the phyB mutant, and FT expression was repressed in the voz1 voz2 mutant. Green fluorescent protein-VOZ2 signal was observed in the cytoplasm, and interaction of VOZ proteins with phyB was indicated to occur in the cytoplasm under far-red light. However, VOZ2 protein modified to localize constitutively in the nucleus promoted flowering. In addition, the stability of VOZ2 proteins in the nucleus was modulated by light quality in a phytochrome-dependent manner. We propose that partial translocation of VOZ proteins from the cytoplasm to the nucleus mediates the initial step of the phyB signal transduction pathway that regulates flowering.
Figures





References
-
- Al-Sady B., Ni W., Kircher S., Schäfer E., Quail P.H. (2006). Photoactivated phytochrome induces rapid PIF3 phosphorylation prior to proteasome-mediated degradation. Mol. Cell 23: 439–446 - PubMed
-
- An H., Roussot C., Suárez-López P., Corbesier L., Vincent C., Piñeiro M., Hepworth S., Mouradov A., Justin S., Turnbull C., Coupland G. (2004). CONSTANS acts in the phloem to regulate a systemic signal that induces photoperiodic flowering of Arabidopsis. Development 131: 3615–3626 - PubMed
-
- Bäckström S., Elfving N., Nilsson R., Wingsle G., Björklund S. (2007). Purification of a plant mediator from Arabidopsis thaliana identifies PFT1 as the Med25 subunit. Mol. Cell 26: 717–729 - PubMed
-
- Bastow R., Mylne J.S., Lister C., Lippman Z., Martienssen R.A., Dean C. (2004). Vernalization requires epigenetic silencing of FLC by histone methylation. Nature 427: 164–167 - PubMed
-
- Bäurle I., Dean C. (2006). The timing of developmental transitions in plants. Cell 125: 655–664 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

