Progressive vaccinia: case description and laboratory-guided therapy with vaccinia immune globulin, ST-246, and CMX001
- PMID: 22904336
- PMCID: PMC3529603
- DOI: 10.1093/infdis/jis510
Progressive vaccinia: case description and laboratory-guided therapy with vaccinia immune globulin, ST-246, and CMX001
Abstract
Progressive vaccinia (PV) is a rare but potentially lethal complication that develops in smallpox vaccine recipients with severely impaired cellular immunity. We describe a patient with PV who required treatment with vaccinia immune globulin and who received 2 investigational agents, ST-246 and CMX001. We describe the various molecular, pharmacokinetic, and immunologic studies that provided guidance to escalate and then successfully discontinue therapy. Despite development of resistance to ST-246 during treatment, the patient had resolution of PV. This case demonstrates the need for continued development of novel anti-orthopoxvirus pharmaceuticals and the importance of both intensive and timely clinical and laboratory support in management of PV.
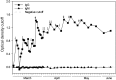
Figures




References
-
- World Health Organization. Declaration of global eradication of smallpox. Wkly Epidemiol Rec. 1980;55:145–52.
-
- Henderson DA, Inglesby TV, Bartlett JG, et al. Smallpox as a biological weapon: medical and public health management. Working Group on Civilian Biodefense. JAMA. 1999;281:2127–37. - PubMed
-
- Reynolds MG, Yorita KL, Kuehnert MJ, et al. Clinical manifestations of human monkeypox influenced by route of infection. J Infect Dis. 2006;194:773–80. - PubMed
-
- Hammarlund E, Lewis MW, Hansen SG, et al. Duration of antiviral immunity after smallpox vaccination. Nat Med. 2003;9:1131–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

