Real time identification of drug-induced liver injury (DILI) through daily screening of ALT results: a prospective pilot cohort study
- PMID: 22905129
- PMCID: PMC3419230
- DOI: 10.1371/journal.pone.0042418
Real time identification of drug-induced liver injury (DILI) through daily screening of ALT results: a prospective pilot cohort study
Abstract
Objective: Identification of drug-induced liver disease (DILI) is difficult, even among hospitalized patients. The aim of this pilot study was to assess the impact of a specific strategy for DILI screening.
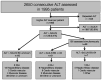
Design: We prospectively compared the number of acute DILI cases identified in one week of a proactive strategy based on centralized elevated ALT values to those identified with a standard of care strategy for 24-week period based on referral cases to the hepatology unit. In the centralized strategy, a designated study biochemist identified patients with ALT greater than 3 times the upper limit of normal values (ULN) and notified the designated hepatologists, who then went to the patients' wards, analyzed the charts, and if necessary, interviewed the identified patients. During these two periods, patients with possible DILI were included after signing an informed consent in an ongoing European diagnostic study (SAFE-T consortium).
Results: During the 24-week period of the standard strategy, 12 (0.04%) patients out of a total of 28,145 were identified as having possible DILI, and 11 of these accepted to be included in the protocol. During the one-week proactive period, 7 patients out of a total of 1407 inpatients (0.498%) [odds ratio vs. standard = 12.1 (95% CI, 3.9-32.3); P<0.0001] were identified with possible DILI, and 5 were included in the protocol.
Conclusion: A simple strategy based on the daily analysis of cases with ALT >3 ULN by designated biochemists and hepatologists identified 12 times more acute cases of drug-induced liver disease than the standard strategy. This pilot cohort is registered on the number AP-HP P110201/1/08-03-2011 and AFSSAPS B110346-70.
Conflict of interest statement
Figures
References
-
- Lazarou J, Pomeranz BH, Corey PN (1998) Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 279: 1200–1205. - PubMed
-
- Verma S, Kaplowitz N (2009) Diagnosis, management and prevention of drug-induced liver injury. Gut 58: 1555–1564. - PubMed
-
- Meier Y, Cavallaro M, Roos M, Pauli-Magnus C, Folkers G, et al. (2005) Incidence of drug-induced liver injury in medical inpatients. Eur J Clin Pharmaco l61: 135–143. - PubMed
-
- Watkins PB, Seligman PJ, Pears JS, Avigan MI, Senior JR (2008) Using controlled clinical trials to learn more about acute drug-induced liver injury. Hepatology 48: 1680–1689. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous