Novel virostatic agents against bluetongue virus
- PMID: 22905259
- PMCID: PMC3419696
- DOI: 10.1371/journal.pone.0043341
Novel virostatic agents against bluetongue virus
Abstract
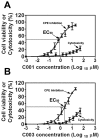
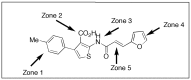
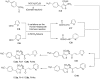
Bluetongue virus (BTV), a member in the family Reoviridae, is a re-emerging animal disease infecting cattle and sheep. With its recent outbreaks in Europe, there is a pressing need for efficacious antivirals. We presented here the identification and characterization of a novel virostatic molecule against BTV, an aminothiophenecarboxylic acid derivative named compound 003 (C003). The virostatic efficacy of C003 could be improved via chemical modification, leading to a de novo synthesized compound 052 (C052). The 50% effective concentrations (EC(50)) of C003 and C052 were determined at 1.76 ± 0.73 µM and 0.27 ± 0.12 µM, respectively. The 50% cytotoxicity concentration (CC(50)) of C003 was over 100 µM and the CC(50) of C052 was at 82.69 µM. Accordingly, the 50% selective index (SI(50)) of C003 and C052 against BTV was over 57 and 306, respectively. The inhibitory effect of C003/C052 on BTV-induced apoptosis was also confirmed via the inhibition of caspase-3/-7 activation post BTV infection. C003/C052 could inhibit BTV induced CPE even when added as late as 24 h.p.i., indicating that they might act at late stage of viral life-cycle. C003/C052 could reduce over two-logs of both the progeny virus production and the number of genomic viral RNA copies. Interestingly, both the activation of host autophagy and viral protein expression were inhibited post BTV infection when cells were treated with C003 and C052, suggesting that C003/C052 might act as virostatic agents via inhibiting host autophagy activation. Although further investigations might be needed to pin down the exact mechanism of C003/C052, our finding suggested that these compounds might be potent lead compounds with potential novel mechanism of action against BTV.
Conflict of interest statement
Figures


Similar articles
-
Assays for the identification of novel antivirals against bluetongue virus.J Vis Exp. 2013 Oct 11;(80):50820. doi: 10.3791/50820. J Vis Exp. 2013. PMID: 24145313 Free PMC article.
-
Requirements and comparative analysis of reverse genetics for bluetongue virus (BTV) and African horse sickness virus (AHSV).Virol J. 2016 Jul 2;13:119. doi: 10.1186/s12985-016-0574-7. Virol J. 2016. PMID: 27368544 Free PMC article.
-
Bluetongue virus without NS3/NS3a expression is not virulent and protects against virulent bluetongue virus challenge.J Gen Virol. 2014 Sep;95(Pt 9):2019-2029. doi: 10.1099/vir.0.065615-0. Epub 2014 Jun 9. J Gen Virol. 2014. PMID: 24914064
-
Recombinant vaccines against bluetongue virus.Virus Res. 2014 Mar;182:78-86. doi: 10.1016/j.virusres.2013.11.013. Epub 2013 Nov 25. Virus Res. 2014. PMID: 24287057 Review.
-
An updated review on bluetongue virus: epidemiology, pathobiology, and advances in diagnosis and control with special reference to India.Vet Q. 2020 Dec;40(1):258-321. doi: 10.1080/01652176.2020.1831708. Vet Q. 2020. PMID: 33003985 Free PMC article. Review.
Cited by
-
Antagonistic effects of lycopene on cadmium-induced hippocampal dysfunctions in autophagy, calcium homeostatis and redox.Oncotarget. 2017 Jul 4;8(27):44720-44731. doi: 10.18632/oncotarget.18249. Oncotarget. 2017. PMID: 28615536 Free PMC article.
-
Epizootic hemorrhagic disease virus induces and benefits from cell stress, autophagy, and apoptosis.J Virol. 2013 Dec;87(24):13397-408. doi: 10.1128/JVI.02116-13. Epub 2013 Oct 2. J Virol. 2013. PMID: 24089565 Free PMC article.
-
Inhibition of Orbivirus Replication by Fluvastatin and Identification of the Key Elements of the Mevalonate Pathway Involved.Viruses. 2021 Jul 23;13(8):1437. doi: 10.3390/v13081437. Viruses. 2021. PMID: 34452303 Free PMC article.
-
Whole-transcriptome analyses of sheep embryonic testicular cells infected with the bluetongue virus.Front Immunol. 2022 Dec 1;13:1053059. doi: 10.3389/fimmu.2022.1053059. eCollection 2022. Front Immunol. 2022. PMID: 36532076 Free PMC article.
-
Viral RNA intermediates as targets for detection and discovery of novel and emerging mosquito-borne viruses.PLoS Negl Trop Dis. 2015 Mar 23;9(3):e0003629. doi: 10.1371/journal.pntd.0003629. eCollection 2015 Mar. PLoS Negl Trop Dis. 2015. PMID: 25799391 Free PMC article.
References
-
- Li Q, Li H, Blitvich BJ, Zhang J (2007) The Aedes albopictus inhibitor of apoptosis 1 gene protects vertebrate cells from bluetongue virus-induced apoptosis. Insect Mol Biol 16: 93–105. - PubMed
-
- Schwartz-Cornil I, Mertens PP, Contreras V, Hemati B, Pascale F, et al. (2008) Bluetongue virus: virology, pathogenesis and immunity. Vet Res 39: 46. - PubMed
-
- Mellor PS, Wittmann EJ (2002) Bluetongue virus in the Mediterranean Basin 1998–2001. Vet J 164: 20–37. - PubMed
-
- Paweska JT, Venter GJ, Mellor PS (2002) Vector competence of South African Culicoides species for bluetongue virus serotype 1 (BTV-1) with special reference to the effect of temperature on the rate of virus replication in C. imicola and C. bolitinos. Med Vet Entomol 16: 10–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

