Population pharmacokinetics of piperacillin at two dose levels: influence of nonlinear pharmacokinetics on the pharmacodynamic profile
- PMID: 22908169
- PMCID: PMC3486597
- DOI: 10.1128/AAC.00937-12
Population pharmacokinetics of piperacillin at two dose levels: influence of nonlinear pharmacokinetics on the pharmacodynamic profile
Abstract
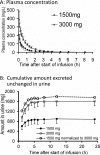
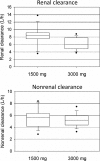
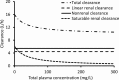
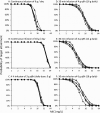
Piperacillin in combination with tazobactam is one of the most commonly used intravenous antibiotics. There is evidence for a possible saturable elimination of piperacillin. Therefore, the saturable elimination and its impact on the choice of optimal dosage regimens were quantified. In a randomized crossover study, 10 healthy volunteers received 1,500 mg and 3,000 mg of piperacillin as 5-min intravenous infusion. Population pharmacokinetics based on plasma and urine data were determined utilizing NONMEM and S-ADAPT. Probabilities of target attainment (PTAs) were compared for different models and dosage regimens, based on the target time of the non-protein-bound concentration above the MIC of at least 50% of the dosing interval. Total clearance of piperacillin was 18% (geometric mean ratio, 90% confidence interval, 11 to 24%) lower (P < 0.01), and renal clearance was 24% (9 to 37%) lower (P = 0.02) at the high compared to the low dose. The final model included first-order nonrenal elimination and parallel first-order and mixed-order renal elimination. Nonrenal clearance was 5.44 liter/h (coefficient of variation, 18%), first-order renal clearance was 4.42 liter/h (47%), and the maximum elimination rate of mixed-order renal elimination was 219 mg/h (84%), with a Michaelis-Menten constant of 36.1 mg/liter (112%). Compared to models with saturable elimination, a linear model predicted up to 10% lower population PTAs for high-dose short-term infusions (6 g every 8 h) and up to 4% higher population PTAs for low-dose continuous infusions (6 g/day). While renal elimination of piperacillin was saturable at therapeutic concentrations, the extent of saturation of nonrenal clearance was small. The influence of saturable elimination on PTAs for clinically relevant dosage regimens was relatively small.
Figures

Similar articles
-
Population Pharmacokinetics and Pharmacodynamics of Extended-Infusion Piperacillin and Tazobactam in Critically Ill Children.Antimicrob Agents Chemother. 2015 Nov 9;60(1):522-31. doi: 10.1128/AAC.02089-15. Print 2016 Jan. Antimicrob Agents Chemother. 2015. PMID: 26552978 Free PMC article.
-
Nonlinear pharmacokinetics of piperacillin in healthy volunteers--implications for optimal dosage regimens.Br J Clin Pharmacol. 2010 Nov;70(5):682-93. doi: 10.1111/j.1365-2125.2010.03750.x. Br J Clin Pharmacol. 2010. PMID: 21039762 Free PMC article.
-
Population pharmacokinetics of extended-infusion piperacillin-tazobactam in hospitalized patients with nosocomial infections.Antimicrob Agents Chemother. 2012 Aug;56(8):4087-94. doi: 10.1128/AAC.00521-12. Epub 2012 May 14. Antimicrob Agents Chemother. 2012. PMID: 22585219 Free PMC article.
-
Comparative pharmacodynamics of intermittent and prolonged infusions of piperacillin/tazobactam using Monte Carlo simulation and steady-state pharmacokinetic data from hospitalized patients.Ann Pharmacother. 2009 Nov;43(11):1887-9. doi: 10.1345/aph.1M462. Epub 2009 Oct 6. Ann Pharmacother. 2009. PMID: 19809005 Review.
-
Pharmacokinetic evaluation of piperacillin-tazobactam.Expert Opin Drug Metab Toxicol. 2010 Aug;6(8):1017-31. doi: 10.1517/17425255.2010.506187. Expert Opin Drug Metab Toxicol. 2010. PMID: 20636224 Review.
Cited by
-
Piperacillin-Tazobactam in Intensive Care Units: A Review of Population Pharmacokinetic Analyses.Clin Pharmacokinet. 2021 Jul;60(7):855-875. doi: 10.1007/s40262-021-01013-1. Epub 2021 Apr 20. Clin Pharmacokinet. 2021. PMID: 33876381 Review.
-
A Pooled Pharmacokinetic Analysis for Piperacillin/Tazobactam Across Different Patient Populations: From Premature Infants to the Elderly.Clin Pharmacokinet. 2025 Jan;64(1):107-126. doi: 10.1007/s40262-024-01460-6. Epub 2024 Dec 25. Clin Pharmacokinet. 2025. PMID: 39722108 Free PMC article.
-
Personalized Antibiotic Therapy for the Critically Ill: Implementation Strategies and Effects on Clinical Outcome of Piperacillin Therapeutic Drug Monitoring-A Descriptive Retrospective Analysis.Antibiotics (Basel). 2021 Nov 26;10(12):1452. doi: 10.3390/antibiotics10121452. Antibiotics (Basel). 2021. PMID: 34943664 Free PMC article.
-
Antibiotics in Adult Cystic Fibrosis Patients: A Review of Population Pharmacokinetic Analyses.Clin Pharmacokinet. 2021 Apr;60(4):447-470. doi: 10.1007/s40262-020-00970-3. Epub 2021 Jan 15. Clin Pharmacokinet. 2021. PMID: 33447944
-
Pharmacodynamic model for β-lactam regimens used in surgical prophylaxis: model-based evaluation of standard dosing regimens.Int J Clin Pharm. 2018 Oct;40(5):1059-1071. doi: 10.1007/s11096-018-0720-y. Epub 2018 Aug 16. Int J Clin Pharm. 2018. PMID: 30117081
References
-
- Ambrose PG, Bhavnani SM, Jones RN. 2003. Pharmacokinetics-pharmacodynamics of cefepime and piperacillin-tazobactam against Escherichia coli and Klebsiella pneumoniae strains producing extended-spectrum beta-lactamases: report from the ARREST program. Antimicrob. Agents Chemother. 47:1643–1646 - PMC - PubMed
-
- Ambrose PG, et al. 2007. Pharmacokinetics-pharmacodynamics of antimicrobial therapy: it's not just for mice anymore. Clin. Infect. Dis. 44:79–86 - PubMed
-
- Andes D, Craig WA. 2002. Animal model pharmacokinetics and pharmacodynamics: a critical review. Int. J. Antimicrob. Agents 19:261–268 - PubMed
-
- Anonymous 1999. Piperacillin sodium and tazobactam sodium (Zosyn) product information. Lederle Laboratories, Pearl River, NY
-
- Aronoff GR, Sloan RS, Brier ME, Luft FC. 1983. The effect of piperacillin dose on elimination kinetics in renal impairment. Eur. J. Clin. Pharmacol. 24:543–547 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

