Glycosylation is dispensable for sorting of synaptotagmin 1 but is critical for targeting of SV2 and synaptophysin to recycling synaptic vesicles
- PMID: 22908222
- PMCID: PMC3471705
- DOI: 10.1074/jbc.M112.398883
Glycosylation is dispensable for sorting of synaptotagmin 1 but is critical for targeting of SV2 and synaptophysin to recycling synaptic vesicles
Abstract
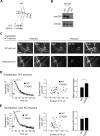
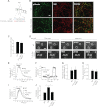
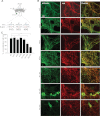
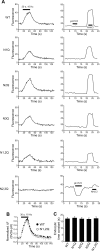
Glycosylation is a major form of post-translational modification of synaptic vesicle membrane proteins. For example, the three major synaptic vesicle glycoproteins, synaptotagmin 1, synaptophysin, and SV2, represent ∼30% of the total copy number of vesicle proteins. Previous studies suggested that glycosylation is required for the vesicular targeting of synaptotagmin 1, but the role of glycosylation of synaptophysin and SV2 has not been explored in detail. In this study, we analyzed all glycosylation sites on synaptotagmin 1, synaptophysin, and SV2A via mutagenesis and optical imaging of pHluorin-tagged proteins in cultured neurons from knock-out mice lacking each protein. Surprisingly, these experiments revealed that glycosylation is completely dispensable for the sorting of synaptotagmin 1 to SVs whereas the N-glycans on SV2A are only partially dispensable. In contrast, N-glycan addition is essential for the synaptic localization and function of synaptophysin. Thus, glycosylation plays distinct roles in the trafficking of each of the three major synaptic vesicle glycoproteins.
Figures


References
-
- Bonanomi D., Benfenati F., Valtorta F. (2006) Protein sorting in the synaptic vesicle life cycle. Prog. Neurobiol. 80, 177–217 - PubMed
-
- Hannah M. J., Schmidt A. A., Huttner W. B. (1999) Synaptic vesicle biogenesis. Annu. Rev. Cell Dev. Biol. 15, 733–798 - PubMed
-
- Sampo B., Kaech S., Kunz S., Banker G. (2003) Two distinct mechanisms target membrane proteins to the axonal surface. Neuron 37, 611–624 - PubMed
-
- Kittler J. T., Moss S. J. (2003) Modulation of GABAA receptor activity by phosphorylation and receptor trafficking: implications for the efficacy of synaptic inhibition. Curr. Opin. Neurobiol. 13, 341–347 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

