Blocking antibody to the β-subunit of FSH prevents bone loss by inhibiting bone resorption and stimulating bone synthesis
- PMID: 22908268
- PMCID: PMC3437842
- DOI: 10.1073/pnas.1212806109
Blocking antibody to the β-subunit of FSH prevents bone loss by inhibiting bone resorption and stimulating bone synthesis
Abstract
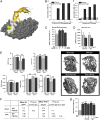
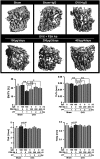
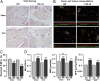
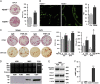
Low estrogen levels undoubtedly underlie menopausal bone thinning. However, rapid and profuse bone loss begins 3 y before the last menstrual period, when serum estrogen is relatively normal. We have shown that the pituitary hormone FSH, the levels of which are high during late perimenopause, directly stimulates bone resorption by osteoclasts. Here, we generated and characterized a polyclonal antibody to a 13-amino-acid-long peptide sequence within the receptor-binding domain of the FSH β-subunit. We show that the FSH antibody binds FSH specifically and blocks its action on osteoclast formation in vitro. When injected into ovariectomized mice, the FSH antibody attenuates bone loss significantly not only by inhibiting bone resorption, but also by stimulating bone formation, a yet uncharacterized action of FSH that we report herein. Mesenchymal cells isolated from mice treated with the FSH antibody show greater osteoblast precursor colony counts, similarly to mesenchymal cells isolated from FSH receptor (FSHR)(-/-) mice. This suggests that FSH negatively regulates osteoblast number. We confirm that this action is mediated by signaling-efficient FSHRs present on mesenchymal stem cells. Overall, the data prompt the future development of an FSH-blocking agent as a means of uncoupling bone formation and bone resorption to a therapeutic advantage in humans.
Conflict of interest statement
Conflict of interest statement: M.Z. is a named inventor of a pending patent application related to osteoclastic bone resorption filed by the Mount Sinai School of Medicine (MSSM). In the event the pending or issued patent is licensed, he would be entitled to a share of any proceeds MSSM receives from the licensee.
Figures
References
-
- Randolph JF, Jr, et al. Change in estradiol and follicle-stimulating hormone across the early menopausal transition: Effects of ethnicity and age. J Clin Endocrinol Metab. 2004;89:1555–1561. - PubMed
-
- Sowers MR, et al. Hormone predictors of bone mineral density changes during the menopausal transition. J Clin Endocrinol Metab. 2006;91:1261–1267. - PubMed
-
- Zaidi M. Skeletal remodeling in health and disease. Nat Med. 2007;13:791–801. - PubMed
-
- Pazianas M, Epstein S, Zaidi M. Evaluating the antifracture efficacy of bisphosphonates. Rev Recent Clin Trials. 2009;4:122–130. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

