The Ndc80 kinetochore complex directly modulates microtubule dynamics
- PMID: 22908300
- PMCID: PMC3479545
- DOI: 10.1073/pnas.1209615109
The Ndc80 kinetochore complex directly modulates microtubule dynamics
Abstract
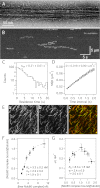
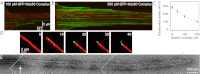
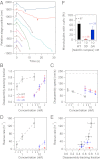
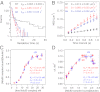
The conserved Ndc80 complex is an essential microtubule-binding component of the kinetochore. Recent findings suggest that the Ndc80 complex influences microtubule dynamics at kinetochores in vivo. However, it was unclear if the Ndc80 complex mediates these effects directly, or by affecting other factors localized at the kinetochore. Using a reconstituted system in vitro, we show that the human Ndc80 complex directly stabilizes the tips of disassembling microtubules and promotes rescue (the transition from microtubule shortening to growth). In vivo, an N-terminal domain in the Ndc80 complex is phosphorylated by the Aurora B kinase. Mutations that mimic phosphorylation of the Ndc80 complex prevent stable kinetochore-microtubule attachment, and mutations that block phosphorylation damp kinetochore oscillations. We find that the Ndc80 complex with Aurora B phosphomimetic mutations is defective at promoting microtubule rescue, even when robustly coupled to disassembling microtubule tips. This impaired ability to affect dynamics is not simply because of weakened microtubule binding, as an N-terminally truncated complex with similar binding affinity is able to promote rescue. Taken together, these results suggest that in addition to regulating attachment stability, Aurora B controls microtubule dynamics through phosphorylation of the Ndc80 complex.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Comment in
-
Complications dawn for kinetochore regulation by Aurora.Proc Natl Acad Sci U S A. 2012 Oct 2;109(40):15972-3. doi: 10.1073/pnas.1214115109. Epub 2012 Sep 18. Proc Natl Acad Sci U S A. 2012. PMID: 22991465 Free PMC article. No abstract available.
Similar articles
-
The Ndc80 complex uses a tripartite attachment point to couple microtubule depolymerization to chromosome movement.Mol Biol Cell. 2011 Apr 15;22(8):1217-26. doi: 10.1091/mbc.E10-07-0626. Epub 2011 Feb 16. Mol Biol Cell. 2011. PMID: 21325630 Free PMC article.
-
Phosphorylation of HsMis13 by Aurora B kinase is essential for assembly of functional kinetochore.J Biol Chem. 2008 Sep 26;283(39):26726-36. doi: 10.1074/jbc.M804207200. Epub 2008 Jul 17. J Biol Chem. 2008. PMID: 18640974 Free PMC article.
-
Dynamic acetylation of the kinetochore-associated protein HEC1 ensures accurate microtubule-kinetochore attachment.J Biol Chem. 2019 Jan 11;294(2):576-592. doi: 10.1074/jbc.RA118.003844. Epub 2018 Nov 8. J Biol Chem. 2019. PMID: 30409912 Free PMC article.
-
The Molecular Mechanism of Aurora-B Regulating Kinetochore-Microtubule Attachment in Mitosis and Oocyte Meiosis.Cytogenet Genome Res. 2024;164(2):69-77. doi: 10.1159/000540588. Epub 2024 Jul 27. Cytogenet Genome Res. 2024. PMID: 39068909 Review.
-
Detection and correction of merotelic kinetochore orientation by Aurora B and its partners.Cell Cycle. 2007 Jul 1;6(13):1558-64. doi: 10.4161/cc.6.13.4452. Epub 2007 May 18. Cell Cycle. 2007. PMID: 17603301 Review.
Cited by
-
Complications dawn for kinetochore regulation by Aurora.Proc Natl Acad Sci U S A. 2012 Oct 2;109(40):15972-3. doi: 10.1073/pnas.1214115109. Epub 2012 Sep 18. Proc Natl Acad Sci U S A. 2012. PMID: 22991465 Free PMC article. No abstract available.
-
Spatiotemporal organization of branched microtubule networks.Elife. 2019 May 8;8:e43890. doi: 10.7554/eLife.43890. Elife. 2019. PMID: 31066674 Free PMC article.
-
Kinetochores require oligomerization of Dam1 complex to maintain microtubule attachments against tension and promote biorientation.Nat Commun. 2014 Sep 19;5:4951. doi: 10.1038/ncomms5951. Nat Commun. 2014. PMID: 25236177 Free PMC article.
-
Human Ska complex and Ndc80 complex interact to form a load-bearing assembly that strengthens kinetochore-microtubule attachments.Proc Natl Acad Sci U S A. 2018 Mar 13;115(11):2740-2745. doi: 10.1073/pnas.1718553115. Epub 2018 Feb 27. Proc Natl Acad Sci U S A. 2018. PMID: 29487209 Free PMC article.
-
Coregulation of NDC80 Complex Subunits Determines the Fidelity of the Spindle-Assembly Checkpoint and Mitosis.Mol Cancer Res. 2024 May 2;22(5):423-439. doi: 10.1158/1541-7786.MCR-23-0828. Mol Cancer Res. 2024. PMID: 38324016 Free PMC article.
References
-
- Liu D, Lampson MA. Regulation of kinetochore-microtubule attachments by Aurora B kinase. Biochem Soc Trans. 2009;37:976–980. - PubMed
-
- Cheeseman IM, et al. Phospho-regulation of kinetochore-microtubule attachments by the Aurora kinase Ipl1p. Cell. 2002;111:163–172. - PubMed
-
- Pinsky BA, Kung C, Shokat KM, Biggins S. The Ipl1-Aurora protein kinase activates the spindle checkpoint by creating unattached kinetochores. Nat Cell Biol. 2006;8:78–83. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

