Effects of activation on functional aster formation, microtubule assembly, and blastocyst development of goat oocytes injected with round spermatids
- PMID: 22908906
- PMCID: PMC3459009
- DOI: 10.1089/cell.2012.0029
Effects of activation on functional aster formation, microtubule assembly, and blastocyst development of goat oocytes injected with round spermatids
Abstract
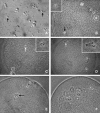
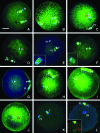
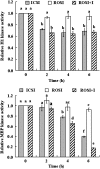
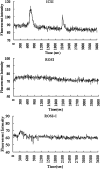
A systematic study was conducted on round spermatids (ROS) injection (ROSI) using the goat model. After ROSI, the oocytes were treated or not with ionomycin (ROSI+I and ROSI-I, respectively) and compared with intracytoplasmic sperm injection (ICSI). After ROSI-I, most oocytes were arrested with premature chromatin condensation and few oocytes formed pronuclei. In contrast, most oocytes formed pronuclei after ROSI+I. Some ROS were observed to form asters that organized a dense microtubule network after ROSI+I, but after ROSI-I, no ROS asters were observed. Whereas most of the oocytes showed Ca(2+) rises and a significant decline in maturation-promoting factor (MPF) and mitogen-activated protein kinase (MAPK) activities after ROSI+I, no such changes were observed after ROSI-I. Due to the lack of Ca(2+) oscillations after ROSI-I, oocytes were injected with more ROS. Interestingly, different from the results observed in a single ROS injection, injection with four ROS effectively activated oocytes by inducing typical Ca(2+) oscillations. Whereas ROSI+I oocytes and ICSI oocytes both showed extensive microtubule networks, no such a network was observed in parthenogenetic oocytes. Together, the results suggest that goat ROS is not able to trigger intracellular Ca(2+) rises and thus to inhibit MPF and MAPK activities, but artificial activation improved fertilization and development of ROSI goat oocytes. Goat ROS can organize functional microtubular asters in activated oocytes. A ROS-derived factor(s) may be essential for organization of a functional microtubule network to unite pronuclei. Goat centrosome is of paternal origin because both ROS and sperm asters organized an extensive microtubule network after intra-oocyte injection.
Figures
Similar articles
-
Functional assessment of centrosomes of spermatozoa and spermatids microinjected into rabbit oocytes.Mol Reprod Dev. 2009 Mar;76(3):270-7. doi: 10.1002/mrd.20951. Mol Reprod Dev. 2009. PMID: 18646048
-
Similar time restriction for intracytoplasmic sperm injection and round spermatid injection into activated oocytes for efficient offspring production.Biol Reprod. 2004 Jun;70(6):1863-9. doi: 10.1095/biolreprod.103.025171. Epub 2004 Feb 25. Biol Reprod. 2004. PMID: 14985245
-
Activation regimens for full-term development of rabbit oocytes injected with round spermatids.Mol Reprod Dev. 2009 Jun;76(6):573-9. doi: 10.1002/mrd.20984. Mol Reprod Dev. 2009. PMID: 19062169
-
Fertilization and developmental initiation of oocytes by injection of spermatozoa and pre-spermatozoal cells.Ital J Anat Embryol. 2005;110(2 Suppl 1):145-50. Ital J Anat Embryol. 2005. PMID: 16101032 Review.
-
Oocyte activation after intracytoplasmic injection of mature and immature sperm cells.Hum Reprod. 1998 Apr;13 Suppl 1:117-27. doi: 10.1093/humrep/13.suppl_1.117. Hum Reprod. 1998. PMID: 9663776 Review.
Cited by
-
The Role of Sperm Centrioles in Human Reproduction - The Known and the Unknown.Front Cell Dev Biol. 2019 Oct 1;7:188. doi: 10.3389/fcell.2019.00188. eCollection 2019. Front Cell Dev Biol. 2019. PMID: 31632960 Free PMC article. Review.
References
-
- Ajduk A. Małagocki A. Maleszewski M. Cytoplasmic maturation of mammalian oocytes: Development of a mechanism responsible for sperm-induced Ca2+ oscillations. Reprod. Biol. 2008;8:3–22. - PubMed
-
- Al-Hasani S. Ludwig M. Palermo I. Küpker W. Sandmann J. Johannisson R. Fornara P. Sturm R. Bals-Pratsch M. Bauer O. Diedrich K. Intracytoplasmic injection of round and elongated spermatids from azoospermic patients: results and review. Hum. Reprod. 1999;14(Suppl 1):97–107. - PubMed
-
- Balaban B. Urman B. Isiklar A. Alatas C. Aksoy S. Mercan R. Nuhoglu A. Progression to the blastocyst stage of embryos derived from testicular round spermatids. Hum. Reprod. 2000;15:1377–1382. - PubMed
-
- Chang H.Y. Levasseur M. Jones K.T. Degradation of APCcdc20 and APCcdh1 substrates during the second meiotic division in mouse eggs. J. Cell Sci. 2004;117(Pt 26):6289–6296. - PubMed
-
- Craft I. Bennett V. Nicholson N. Fertilizing ability of testicular spermatozoa. Lancet. 1993;342:864. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

