Ontogeny of the maize shoot apical meristem
- PMID: 22911570
- PMCID: PMC3462627
- DOI: 10.1105/tpc.112.099614
Ontogeny of the maize shoot apical meristem
Abstract
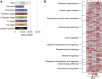
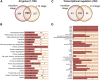
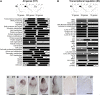
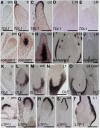
The maize (Zea mays) shoot apical meristem (SAM) arises early in embryogenesis and functions during stem cell maintenance and organogenesis to generate all the aboveground organs of the plant. Despite its integral role in maize shoot development, little is known about the molecular mechanisms of SAM initiation. Laser microdissection of apical domains from developing maize embryos and seedlings was combined with RNA sequencing for transcriptomic analyses of SAM ontogeny. Molecular markers of key events during maize embryogenesis are described, and comprehensive transcriptional data from six stages in maize shoot development are generated. Transcriptomic profiling before and after SAM initiation indicates that organogenesis precedes stem cell maintenance in maize; analyses of the first three lateral organs elaborated from maize embryos provides insight into their homology and to the identity of the single maize cotyledon. Compared with the newly initiated SAM, the mature SAM is enriched for transcripts that function in transcriptional regulation, hormonal signaling, and transport. Comparisons of shoot meristems initiating juvenile leaves, adult leaves, and husk leaves illustrate differences in phase-specific (juvenile versus adult) and meristem-specific (SAM versus lateral meristem) transcript accumulation during maize shoot development. This study provides insight into the molecular genetics of SAM initiation and function in maize.
Figures



Comment in
-
Transcriptome study outlines ontogeny of the maize shoot apical meristem.Plant Cell. 2012 Aug;24(8):3169. doi: 10.1105/tpc.112.240812. Epub 2012 Aug 28. Plant Cell. 2012. PMID: 22932673 Free PMC article. No abstract available.
References
-
- Abbe E.C., Stein O.L. (1954). The growth of the shoot apex in maize: Embryogeny. Am. J. Bot. 41: 285–293
-
- Abedon B.G., Hatfield R.D., Tracy W.F. (2006). Cell wall composition in juvenile and adult leaves of maize (Zea mays L.). J. Agric. Food Chem. 54: 3896–3900 - PubMed
-
- Aida M., Ishida T., Tasaka M. (1999). Shoot apical meristem and cotyledon formation during Arabidopsis embryogenesis: Interaction among the CUP-SHAPED COTYLEDON and SHOOT MERISTEMLESS genes. Development 126: 1563–1570 - PubMed
-
- Boyd L. (1931). Evolution in the monocotyledonous seedling, a new interpretation of the grass embryo. Trans. Bot. Soc. Edinburgh 30: 286–302
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

