The Drosophila MI-2 chromatin-remodeling factor regulates higher-order chromatin structure and cohesin dynamics in vivo
- PMID: 22912596
- PMCID: PMC3415455
- DOI: 10.1371/journal.pgen.1002878
The Drosophila MI-2 chromatin-remodeling factor regulates higher-order chromatin structure and cohesin dynamics in vivo
Abstract
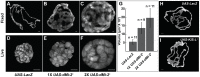
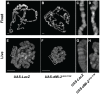
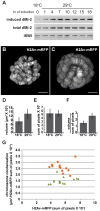
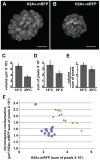
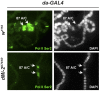
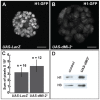
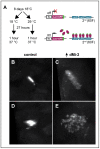
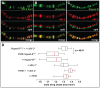
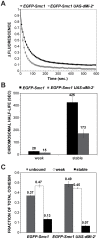
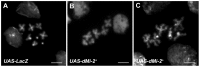
dMi-2 is a highly conserved ATP-dependent chromatin-remodeling factor that regulates transcription and cell fates by altering the structure or positioning of nucleosomes. Here we report an unanticipated role for dMi-2 in the regulation of higher-order chromatin structure in Drosophila. Loss of dMi-2 function causes salivary gland polytene chromosomes to lose their characteristic banding pattern and appear more condensed than normal. Conversely, increased expression of dMi-2 triggers decondensation of polytene chromosomes accompanied by a significant increase in nuclear volume; this effect is relatively rapid and is dependent on the ATPase activity of dMi-2. Live analysis revealed that dMi-2 disrupts interactions between the aligned chromatids of salivary gland polytene chromosomes. dMi-2 and the cohesin complex are enriched at sites of active transcription; fluorescence-recovery after photobleaching (FRAP) assays showed that dMi-2 decreases stable association of cohesin with polytene chromosomes. These findings demonstrate that dMi-2 is an important regulator of both chromosome condensation and cohesin binding in interphase cells.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Li B, Carey M, Workman JL (2007) The role of chromatin during transcription. Cell 128: 707–719. - PubMed
-
- Bassett A, Cooper S, Wu C, Travers A (2009) The folding and unfolding of eukaryotic chromatin. Curr Opin Genet Dev 19: 159–165. - PubMed
-
- Kouzarides T (2007) Chromatin modifications and their function. Cell 128: 693–705. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

