Second pilot trials of the STAR-Liege protocol for tight glycemic control in critically ill patients
- PMID: 22917085
- PMCID: PMC3511234
- DOI: 10.1186/1475-925X-11-58
Second pilot trials of the STAR-Liege protocol for tight glycemic control in critically ill patients
Abstract
Background: Critically ill patients often present increased insulin resistance and stress-induced hyperglycemia. Tight glycemic control aims to reduce blood glucose (BG) levels and variability while ensuring safety from hypoglycemia. This paper presents the results of the second Belgian clinical trial using the customizable STAR framework in a target-to-range control approach. The main objective is reducing measurement frequency while maintaining performance and safety of the glycemic control.
Methods: The STAR-Liege 2 (SL2) protocol targeted the 100-140 mg/dL glycemic band and offered 2-hourly and 3-hourly interventions. Only insulin rates were adjusted, and nutrition inputs were left to the attending clinicians. This protocol restricted the forecasted risk of BG < 90 mg/dL to a 5% level using a stochastic model of insulin sensitivity to assess patient-specific responses to insulin and its future likely variability to optimize insulin interventions. The clinical trial was performed at the Centre Hospitalier Universitaire de Liege and included 9 patients. Results are compared to 24-hour pre-trial and 24-hour post-trial, but also to the results of the first pilot trial performed in Liege, STAR-Liege 1 (SL1). This trial was approved by the Ethics Committee of the Medical Faculty of the University of Liege (Liege, Belgium).
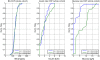
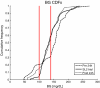
Results: During the SL2 trial, 91 measurements were taken over 194 hours. BG levels were tightly distributed: 54.9% of BG within 100-140 mg/dL, 40.7% were ≥ 140 mg/dL and 4.4% were < 100 mg/dL with no BG < 70 mg/dL. Comparing these results with 24-hour pre-trial and post-trial shows that SL2 reduced high and low BG levels and reduced glycemic variability. Nurses selected 3-hourly measurement only 5 of 16 times and overrode 12% of 91 recommended interventions (35% increased insulin rates and 65% decreased insulin rates). SL1 and SL2 present similar BG levels distribution (p > 0.05) with significantly reduced measurement frequency for SL2 (p < 0.05).
Conclusions: The SL2 protocol succeeded in reducing clinical workload while maintaining safety and effectiveness of the glycemic control. SL2 was also shown to be safer and tighter than hospital control. Overall results validate the efficacy of significantly customizing the STAR framework.
Figures


Similar articles
-
First pilot trial of the STAR-Liege protocol for tight glycemic control in critically ill patients.Comput Methods Programs Biomed. 2012 Nov;108(2):844-59. doi: 10.1016/j.cmpb.2011.07.003. Epub 2011 Aug 31. Comput Methods Programs Biomed. 2012. PMID: 21885150
-
Stochastic targeted (STAR) glycemic control: design, safety, and performance.J Diabetes Sci Technol. 2012 Jan 1;6(1):102-15. doi: 10.1177/193229681200600113. J Diabetes Sci Technol. 2012. PMID: 22401328 Free PMC article.
-
STAR-Liège Clinical Trial Interim Results: Safe and Effective Glycemic Control for All.Annu Int Conf IEEE Eng Med Biol Soc. 2019 Jul;2019:277-280. doi: 10.1109/EMBC.2019.8856303. Annu Int Conf IEEE Eng Med Biol Soc. 2019. PMID: 31945895 Clinical Trial.
-
Glycemic control in the critically ill: What have we learned since NICE-SUGAR?Hosp Pract (1995). 2015;43(3):191-7. doi: 10.1080/21548331.2015.1066227. Hosp Pract (1995). 2015. PMID: 26224425 Review.
-
Intensive insulin therapy in critically ill hospitalized patients: making it safe and effective.J Diabetes Sci Technol. 2011 May 1;5(3):755-67. doi: 10.1177/193229681100500330. J Diabetes Sci Technol. 2011. PMID: 21722591 Free PMC article. Review.
Cited by
-
The benefits of tight glycemic control in critical illness: Sweeter than assumed?Indian J Crit Care Med. 2014 Dec;18(12):807-13. doi: 10.4103/0972-5229.146315. Indian J Crit Care Med. 2014. PMID: 25538415 Free PMC article. Review.
-
Evaluation of an open access software for calculating glucose variability parameters of a continuous glucose monitoring system applied at pediatric intensive care unit.Biomed Eng Online. 2015 Apr 24;14:37. doi: 10.1186/s12938-015-0035-3. Biomed Eng Online. 2015. PMID: 25907677 Free PMC article.
-
Assessment of Glycemic Control Protocol (STAR) Through Compliance Analysis Amongst Malaysian ICU Patients.Med Devices (Auckl). 2020 Jun 4;13:139-149. doi: 10.2147/MDER.S231856. eCollection 2020. Med Devices (Auckl). 2020. PMID: 32607009 Free PMC article.
-
Hypoglycemia Prevention by Algorithm Design During Intravenous Insulin Infusion.Curr Diab Rep. 2018 Mar 26;18(5):26. doi: 10.1007/s11892-018-0994-4. Curr Diab Rep. 2018. PMID: 29582176 Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

