Induction of ligand-specific PrP (C) signaling in human neuronal cells
- PMID: 22918447
- PMCID: PMC3510852
- DOI: 10.4161/pri.21914
Induction of ligand-specific PrP (C) signaling in human neuronal cells
Abstract
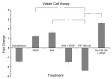
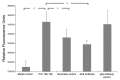
Cellular prion protein (PrP (C) ) has attracted considerable attention for its role in transmissible spongiform encephalopathies (TSEs). In spite of being a point of intense research effort critical questions still remain regarding the physiological function of PrP (C) and how these functions may change with the conversion of the protein into the infectious and pathological conformation (PrP (Sc) ). While emerging evidence suggests PrP (C/Sc) are involved in signal transduction there is little consensus on the signaling pathways associated with the normal and diseased states. The purported involvement of PrP (C) in signal transduction, and the association of TSEs with neural pathology, makes kinome analysis of human neurons an interesting and appropriate model to characterize patterns of signal transduction following activation of PrP (C) by two commonly employed experimental ligands; antibody-induced dimerization by 6H4 and the amino acids 106-126 PrP peptide fragment (PrP 106-126). Analysis of the induced kinome responses reveals distinct patterns of signaling activity following each treatment. Specifically, stimulation of human neurons with the 6H4 antibody results in alterations in mitogen activated protein kinase (MAPK) signaling pathways while the 106-126 peptide activates growth factor related signaling pathways including vascular endothelial growth factor (VEGF) signaling and the phosphoinositide-3 kinase (PI3K) pathway. These pathways were validated through independent functional assays. Collectively these results indicate that stimulation of PrP (C) with distinct ligands, even within the same cell type, results in unique patterns of signaling. While this investigation highlights the apparent functional versatility of PrP (C) as a signaling molecule and may offer insight into cellular mechanisms of TSE pathology it also emphasizes the potential dangers associated with attributing activation of specific intracellular events to particular receptors through artificial models of receptor activation.
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
