The extracellular matrix is a novel attribute of endothelial progenitors and of hypoxic mature endothelial cells
- PMID: 22919069
- PMCID: PMC3509053
- DOI: 10.1096/fj.12-209296
The extracellular matrix is a novel attribute of endothelial progenitors and of hypoxic mature endothelial cells
Abstract
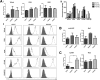
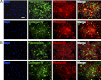
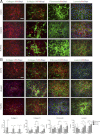
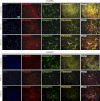
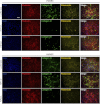
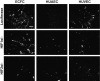
Extracellular matrix (ECM) production is critical to preserve the function and integrity of mature blood vessels. Toward the engineering of blood vessels, studies have centered on ECM production by supporting cells, whereas few studies implicate endothelial cells (ECs) with ECM synthesis. Here, we elucidate variations between cultured human arterial, venous, and progenitor ECs with respect to ECM deposition assembly, composition, and response to biomolecular and physiological factors. Our studies reveal that progenitor ECs, endothelial colony-forming cells (ECFCs), deposit collagen IV, fibronectin, and laminin that assemble to an organized weblike structure, as confirmed by decellularized cultures. Mature ECs only express these ECM proteins intracellularly. ECFC-derived ECM is abrogated in response to TGFβ signaling inhibition and actin cytoskeleton disruption. Hypoxic (1%) and physiological (5%) O(2) tension stimulate ECM deposition from mature ECs. Interestingly, deposition of collagen I is observed only under 5% O(2) tension. ECM production from all ECs is found to be regulated by hypoxia-inducible factors 1α and 2α but differentially in the different cell lines. Collectively, we suggest that ECM deposition and assembly by ECs is dependent on maturation stage and oxygen supply and that these findings can be harnessed to advance engineered vascular therapeutics.
Figures



Similar articles
-
Peroxidasin is essential for endothelial cell survival and growth signaling by sulfilimine crosslink-dependent matrix assembly.FASEB J. 2020 Aug;34(8):10228-10241. doi: 10.1096/fj.201902899R. Epub 2020 Jun 16. FASEB J. 2020. PMID: 32543734
-
An Akt/hypoxia-inducible factor-1alpha/platelet-derived growth factor-BB autocrine loop mediates hypoxia-induced chemoresistance in liver cancer cells and tumorigenic hepatic progenitor cells.Clin Cancer Res. 2009 May 15;15(10):3462-71. doi: 10.1158/1078-0432.CCR-08-2127. Epub 2009 May 15. Clin Cancer Res. 2009. PMID: 19447872
-
Breast cancer cell-derived matrix supports vascular morphogenesis.Am J Physiol Cell Physiol. 2012 Apr 15;302(8):C1243-56. doi: 10.1152/ajpcell.00011.2012. Epub 2012 Jan 25. Am J Physiol Cell Physiol. 2012. PMID: 22277754 Free PMC article.
-
Implications of fibrotic extracellular matrix in diabetic retinopathy.Exp Biol Med (Maywood). 2022 Jul;247(13):1093-1102. doi: 10.1177/15353702221087175. Epub 2022 Apr 11. Exp Biol Med (Maywood). 2022. PMID: 35410521 Free PMC article. Review.
-
Pathogenic mechanisms and therapeutic implications of extracellular matrix remodelling in cerebral vasospasm.Fluids Barriers CNS. 2023 Nov 4;20(1):81. doi: 10.1186/s12987-023-00483-8. Fluids Barriers CNS. 2023. PMID: 37925414 Free PMC article. Review.
Cited by
-
Hypoxia in Breast Cancer-Scientific Translation to Therapeutic and Diagnostic Clinical Applications.Front Oncol. 2021 Mar 11;11:652266. doi: 10.3389/fonc.2021.652266. eCollection 2021. Front Oncol. 2021. PMID: 33777815 Free PMC article. Review.
-
Uterine vasculature remodeling in human pregnancy involves functional macrochimerism by endothelial colony forming cells of fetal origin.Stem Cells. 2013 Jul;31(7):1363-70. doi: 10.1002/stem.1385. Stem Cells. 2013. PMID: 23554274 Free PMC article.
-
Customizable biomaterials as tools for advanced anti-angiogenic drug discovery.Biomaterials. 2018 Oct;181:53-66. doi: 10.1016/j.biomaterials.2018.07.041. Epub 2018 Jul 26. Biomaterials. 2018. PMID: 30077137 Free PMC article. Review.
-
Hypoxia-inducible hydrogels.Nat Commun. 2014 Jun 9;5:4075. doi: 10.1038/ncomms5075. Nat Commun. 2014. PMID: 24909742 Free PMC article.
-
Heterotypic Tumor Spheroids in Agitation-Based Cultures: A Scaffold-Free Cell Model That Sustains Long-Term Survival of Endothelial Cells.Front Bioeng Biotechnol. 2021 Jun 9;9:649949. doi: 10.3389/fbioe.2021.649949. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34178955 Free PMC article.
References
-
- Takahashi T., Kalka C., Masuda H., Chen D., Silver M., Kearney M., Magner M., Isner J. M., Asahara T. (1999) Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nat. Med. 5, 434–438 - PubMed
-
- Yoder M. C. (2009) Defining human endothelial progenitor cells. J. Thromb. Haemost. 7, 49–52 - PubMed
-
- Asahara T., Kawamoto A., Masuda H. (2011) Concise review: circulating endothelial progenitor cells for vascular medicine. Stem Cells 29, 1650–1655 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

