Releasable layer-by-layer assembly of stabilized lipid nanocapsules on microneedles for enhanced transcutaneous vaccine delivery
- PMID: 22920601
- PMCID: PMC3475723
- DOI: 10.1021/nn302639r
Releasable layer-by-layer assembly of stabilized lipid nanocapsules on microneedles for enhanced transcutaneous vaccine delivery
Abstract
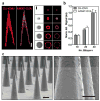
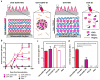
Here we introduce a new approach for transcutaneous drug delivery, using microneedles coated with stabilized lipid nanocapsules, for delivery of a model vaccine formulation. Poly(lactide-co-glycolide) microneedle arrays were coated with multilayer films via layer-by-layer assembly of a biodegradable cationic poly(β-amino ester) (PBAE) and negatively charged interbilayer-cross-linked multilamellar lipid vesicles (ICMVs). To test the potential of these nanocapsule-coated microneedles for vaccine delivery, we loaded ICMVs with a protein antigen and the molecular adjuvant monophosphoryl lipid A. Following application of microneedle arrays to the skin of mice for 5 min, (PBAE/ICMV) films were rapidly transferred from microneedle surfaces into the cutaneous tissue and remained in the skin following removal of the microneedle arrays. Multilayer films implanted in the skin dispersed ICMV cargos in the treated tissue over the course of 24 h in vivo, allowing for uptake of the lipid nanocapsules by antigen presenting cells in the local tissue and triggering their activation in situ. Microneedle-mediated transcutaneous vaccination with ICMV-carrying multilayers promoted robust antigen-specific humoral immune responses with a balanced generation of multiple IgG isotypes, whereas bolus delivery of soluble or vesicle-loaded antigen via intradermal injection or transcutaneous vaccination with microneedles encapsulating soluble protein elicited weak, IgG(1)-biased humoral immune responses. These results highlight the potential of lipid nanocapsules delivered by microneedles as a promising platform for noninvasive vaccine delivery applications.
Figures




Similar articles
-
Intradermal delivery of vaccine nanoparticles using hollow microneedle array generates enhanced and balanced immune response.J Control Release. 2019 Jan 28;294:268-278. doi: 10.1016/j.jconrel.2018.12.026. Epub 2018 Dec 17. J Control Release. 2019. PMID: 30572036
-
Immunostimulatory effect of tetanus toxoid loaded chitosan nanoparticles following microneedles assisted immunization.Nanomedicine. 2016 Jan;12(1):213-22. doi: 10.1016/j.nano.2015.10.009. Epub 2015 Nov 10. Nanomedicine. 2016. PMID: 26554391
-
Skin dendritic cell targeting via microneedle arrays laden with antigen-encapsulated poly-D,L-lactide-co-glycolide nanoparticles induces efficient antitumor and antiviral immune responses.ACS Nano. 2013 Mar 26;7(3):2042-55. doi: 10.1021/nn304235j. Epub 2013 Feb 15. ACS Nano. 2013. PMID: 23373658 Free PMC article.
-
Pharmacokinetic differences between subcutaneous injection and intradermal microneedle delivery of protein therapeutics.Eur J Pharm Biopharm. 2024 Nov;204:114517. doi: 10.1016/j.ejpb.2024.114517. Epub 2024 Sep 28. Eur J Pharm Biopharm. 2024. PMID: 39349073 Review.
-
Microneedle-based drug and vaccine delivery via nanoporous microneedle arrays.Drug Deliv Transl Res. 2015 Aug;5(4):397-406. doi: 10.1007/s13346-015-0238-y. Drug Deliv Transl Res. 2015. PMID: 26044672 Free PMC article. Review.
Cited by
-
Coated and Hollow Microneedle-Mediated Intradermal Immunization in Mice with Diphtheria Toxoid Loaded Mesoporous Silica Nanoparticles.Pharm Res. 2018 Aug 13;35(10):189. doi: 10.1007/s11095-018-2476-4. Pharm Res. 2018. PMID: 30105542 Free PMC article.
-
Nanoparticle-based vaccine delivery for cancer immunotherapy.Immune Netw. 2013 Oct;13(5):177-83. doi: 10.4110/in.2013.13.5.177. Epub 2013 Oct 26. Immune Netw. 2013. PMID: 24198742 Free PMC article. Review.
-
Nanotechnology for protein delivery: Overview and perspectives.J Control Release. 2016 Oct 28;240:24-37. doi: 10.1016/j.jconrel.2015.10.012. Epub 2015 Oct 13. J Control Release. 2016. PMID: 26458789 Free PMC article. Review.
-
Nano-multilamellar lipid vesicles promote the induction of SARS-CoV-2 immune responses by a protein-based vaccine formulation.Nanomedicine. 2022 Sep;45:102595. doi: 10.1016/j.nano.2022.102595. Epub 2022 Aug 27. Nanomedicine. 2022. PMID: 36031045 Free PMC article.
-
Amplify antimicrobial photo dynamic therapy efficacy with poly-beta-amino esters (PBAEs).Sci Rep. 2021 Mar 31;11(1):7275. doi: 10.1038/s41598-021-86773-3. Sci Rep. 2021. PMID: 33790379 Free PMC article.
References
-
- DeVilliers MM, Otto DP, Strydom SJ, Lvov YM. Introduction to Nanocoatings Produced by Layer-by-Layer (LbL) Self-Assembly. Adv Drug Delivery Rev. 2011;63:701–715. - PubMed
-
- Hammond PT. Engineering Materials Layer-by-Layer: Challenges and Apportunities in Multilayer Assembly. AIChE J. 2011;57:2928–2940.
-
- Katagiri K, Hamasaki R, Ariga K, Kikuchi J-i. Layered Paving of Vesicular Nanoparticles Formed with Cerasome as a Bioinspired Organic-Inorganic Hybrid. J Am Chem Soc. 2002;124:7892–7893. - PubMed
-
- Katagiri K, Hamasaki R, Ariga K, Kikuchi J-i. Layer-by-Layer Self-Assembling of Liposomal Nanohybrid “Cerasome” on Substrates. Langmuir. 2002;18:6709–6711.
-
- Graf N, Tanno A, Dochter A, Rothfuchs N, Voeroes J, Zambelli T. Electrochemically Driven delivery to Cells from Vesicles Embedded in Polyelectrolyte Multilayers. Soft Matter. 2012;8:3641–3648.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

