Hsp70 protein complexes as drug targets
- PMID: 22920901
- PMCID: PMC3593251
- DOI: 10.2174/138161213804143699
Hsp70 protein complexes as drug targets
Abstract
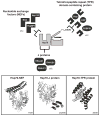
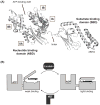
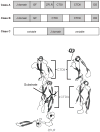
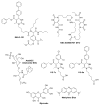
Heat shock protein 70 (Hsp70) plays critical roles in proteostasis and is an emerging target for multiple diseases. However, competitive inhibition of the enzymatic activity of Hsp70 has proven challenging and, in some cases, may not be the most productive way to redirect Hsp70 function. Another approach is to inhibit Hsp70's interactions with important co-chaperones, such as J proteins, nucleotide exchange factors (NEFs) and tetratricopeptide repeat (TPR) domain-containing proteins. These co-chaperones normally bind Hsp70 and guide its many diverse cellular activities. Complexes between Hsp70 and co-chaperones have been shown to have specific functions, including roles in pro-folding, pro-degradation and pro-trafficking pathways. Thus, a promising strategy may be to block protein- protein interactions between Hsp70 and its co-chaperones or to target allosteric sites that disrupt these contacts. Such an approach might shift the balance of Hsp70 complexes and re-shape the proteome and it has the potential to restore healthy proteostasis. In this review, we discuss specific challenges and opportunities related to these goals. By pursuing Hsp70 complexes as drug targets, we might not only develop new leads for therapeutic development, but also discover new chemical probes for use in understanding Hsp70 biology.
Figures

References
-
- Bukau B, Weissman J, Horwich A. Molecular chaperones and protein quality control. Cell. 2006;125:443–51. - PubMed
-
- Frydman J. Folding of newly translated proteins in vivo: the role of molecular chaperones. Ann Rev Biochem. 2001;70:603–47. - PubMed
-
- Hartl FU, Bracher A, Hayer-Hartl M. Molecular chaperones in protein folding and proteostasis. Nature. 2011;475:324–32. - PubMed
-
- Bercovich B, Stancovski I, Mayer A, Blumenfeld N, Laszlo A, Schwartz AL, Ciechanover A. Ubiquitin-dependent degradation of certain protein substrates in vitro requires the molecular chaperone Hsc70. J Biol Chem. 1997;272:9002–10. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

