Ca(2+) influx through L-type Ca(2+) channels and transient receptor potential channels activates pathological hypertrophy signaling
- PMID: 22921230
- PMCID: PMC3472041
- DOI: 10.1016/j.yjmcc.2012.08.005
Ca(2+) influx through L-type Ca(2+) channels and transient receptor potential channels activates pathological hypertrophy signaling
Abstract
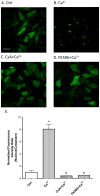
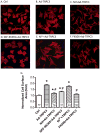
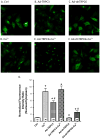
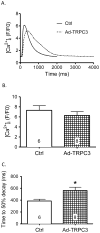
Common cardiovascular diseases such as hypertension and myocardial infarction require that myocytes develop greater than normal force to maintain cardiac pump function. This requires increases in [Ca(2+)]. These diseases induce cardiac hypertrophy and increases in [Ca(2+)] are known to be an essential proximal signal for activation of hypertrophic genes. However, the source of "hypertrophic" [Ca(2+)] is not known and is the topic of this study. The role of Ca(2+) influx through L-type Ca(2+) channels (LTCC), T-type Ca(2+) channels (TTCC) and transient receptor potential (TRP) channels on the activation of calcineurin (Cn)-nuclear factor of activated T cells (NFAT) signaling and myocyte hypertrophy was studied. Neonatal rat ventricular myocytes (NRVMs) and adult feline ventricular myocytes (AFVMs) were infected with an adenovirus containing NFAT-GFP, to determine factors that could induce NFAT nuclear translocation. Four millimolar Ca(2+) or pacing induced NFAT nuclear translocation. This effect was blocked by Cn inhibitors. In NRVMs Nifedipine (Nif, LTCC antagonist) blocked high Ca(2+)-induced NFAT nuclear translocation while SKF-96365 (TRP channel antagonist) and Nickel (Ni, TTCC antagonist) were less effective. The relative potency of these antagonists against Ca(2+) induced NFAT nuclear translocation (Nif>SKF-96365>Ni) was similar to their effects on Ca(2+) transients and the LTCC current. Infection of NRVM with viruses containing TRP channels also activated NFAT-GFP nuclear translocation and caused myocyte hypertrophy. TRP effects were reduced by SKF-96365, but were more effectively antagonized by Nif. These experiments suggest that Ca(2+) influx through LTCCs is the primary source of Ca(2+) to activate Cn-NFAT signaling in NRVMs and AFVMs. While TRP channels cause hypertrophy, they appear to do so through a mechanism involving Ca(2+) entry via LTCCs.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures




References
-
- Bers DM, Guo T. Calcium Signaling in Cardiac Ventricular Myocytes. Annals of the New York Academy of Sciences. 2005;1047:86–98. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

