SAHA, an HDAC inhibitor, synergizes with tacrolimus to prevent murine cardiac allograft rejection
- PMID: 22922441
- PMCID: PMC4002334
- DOI: 10.1038/cmi.2012.28
SAHA, an HDAC inhibitor, synergizes with tacrolimus to prevent murine cardiac allograft rejection
Abstract
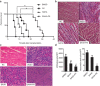
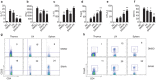
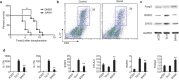
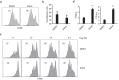
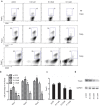
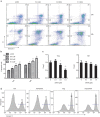
Suberoylanilide hydroxamic acid (SAHA), as a histone deacetylase (HDAC) inhibitor (HDACi), was recently found to exhibit an immunosuppressive effect. However, whether SAHA can synergize with calcineurin inhibitors (CNIs) to inhibit allograft rejection and its underlying mechanism remain elusive. In this study, we demonstrated the synergistic effects of SAHA and non-therapeutic dose of tacrolimus (FK506) in prolonging the allograft survival in a murine cardiac transplant model. Concomitant intragraft examination revealed that allografts from SAHA-treated recipients showed significantly lower levels of IL-17 expression, and no discernable difference for IL-17 expressions was detected between SAHA- and SAHA/FK506-treated allograft as compared with allografts from FK506-treated animals. In contrast, administration of FK506 significantly suppressed interferon (IFN)-γ but increased IL-10 expression as compared with that of SAHA-treated animals, and this effect was independent of SAHA. Interestingly, SAHA synergizes with FK506 to promote Foxp3 and CTLA4 expression. In vitro, SAHA reduced the proportion of Th17 cells in isolated CD4⁺ T-cell population and decreased expressions of IL-17A, IL-17F, STAT3 and RORγt in these cells. Moreover, SAHA enhances suppressive function of regulatory T (Treg) cells by upregulating the expression of CTLA-4 without affecting T effector cell proliferation, and increased the proportion of Treg by selectively promoting apoptosis of T effector cells. Therefore, SAHA, a HDACi, may be a promising immunosuppressive agent with potential benefit in conjunction with CNI drugs.
Figures
References
-
- Nankivell BJ, Kuypers DR. Diagnosis and prevention of chronic kidney allograft loss. Lancet. 2011;378:1428–1437. - PubMed
-
- Hariharan S, Johnson CP, Bresnahan BA, Taranto SE, McIntosh MJ, Stablein D. Improved graft survival after renal transplantation in the United States, 1988 to 1996. N Engl J Med. 2000;342:605–612. - PubMed
-
- Pascual M, Theruvath T, Kawai T, Tolkoff-Rubin N, Cosimi AB. Strategies to improve long-term outcomes after renal transplantation. N Engl J Med. 2002;346:580–590. - PubMed
-
- Casey MJ, Meier-Kriesche HU. Calcineurin inhibitors in kidney transplantation: friend or foe. Curr Opin Nephrol Hypertens. 2011;20:610–615. - PubMed
-
- van de Wetering J, Koumoutsakos P, Peeters A, van der Mast BJ, de Kuiper P, Ijzermans JN, et al. Discontinuation of calcineurin inhibitors treatment allows the development of FOXP3+ regulatory T-cells in patients after kidney transplantation. Clin Transplant. 2011;25:40–46. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

