Glucose transport in Escherichia coli mutant strains with defects in sugar transport systems
- PMID: 22923596
- PMCID: PMC3486086
- DOI: 10.1128/JB.01502-12
Glucose transport in Escherichia coli mutant strains with defects in sugar transport systems
Abstract
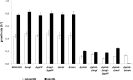
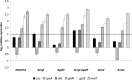
In Escherichia coli, several systems are known to transport glucose into the cytoplasm. The main glucose uptake system under batch conditions is the glucose phosphoenolpyruvate:carbohydrate phosphotransferase system (glucose PTS), but the mannose PTS and the galactose and maltose transporters also can translocate glucose. Mutant strains which lack the enzyme IIBC (EIIBC) protein of the glucose PTS have been investigated previously because their lower rate of acetate formation offers advantages in industrial applications. Nevertheless, a systematic study to analyze the impact of the different glucose uptake systems has not been undertaken. Specifically, how the bacteria cope with the deletion of the major glucose uptake system and which alternative transporters react to compensate for this deficit have not been studied in detail. Therefore, a series of mutant strains were analyzed in aerobic and anaerobic batch cultures, as well as glucose-limited continuous cultivations. Deletion of EIIBC disturbs glucose transport severely in batch cultures; cyclic AMP (cAMP)-cAMP receptor protein (CRP) levels rise, and induction of the mgl operon occurs. Nevertheless, Mgl activity is not essential for growth of these mutants, since deletion of this transporter did not affect the growth rate; the activities of the remaining transporters seem to be sufficient. Under conditions of glucose limitation, mgl is upregulated 23-fold compared to levels for growth under glucose excess. Despite the strong induction of mgl upon glucose limitation, deletion of this transport system did not lead to further changes. Although the galactose transporters are often regarded as important for glucose uptake at micromolar concentrations, the glucose as well as mannose PTS might be sufficient for growth at this relatively low dilution rate.
Figures






References
-
- Bettenbrock K, et al. 2006. A quantitative approach to catabolite repression in Escherichia coli. J. Biol. Chem. 281: 2578–2584 - PubMed
-
- Chen RZ, Yap WMGJ, Postma PW, Bailey JE. 1997. Comparative studies of Escherichia coli strains using different glucose uptake systems: metabolism and energetics. Biotechnol. Bioeng. 56: 583–590 - PubMed
-
- Chou CH, Bennett GN, San KY. 1994. Effect of modified glucose uptake using genetic engineering techniques on high-level recombinant protein production in Escherichia coli dense cultures. Biotechnol. Bioeng. 44: 952–960 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

