Relieved residual damage in the hematopoietic system of mice rescued by radiation-induced adaptive response (Yonezawa Effect)
- PMID: 22923746
- PMCID: PMC3534278
- DOI: 10.1093/jrr/rrs077
Relieved residual damage in the hematopoietic system of mice rescued by radiation-induced adaptive response (Yonezawa Effect)
Abstract
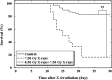
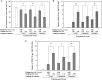
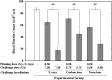
Existence of adaptive response (AR) was previously demonstrated in C57BL/6J mice. Irradiations were performed by delivering a priming low dose of X-rays (0.50 Gy) in combination with a challenge high dose of accelerated carbon or neon ion particles. AR was characterized by significantly decreased mortality in the 30-day survival test. This mouse AR model ('Yonezawa Effect') was originally established by using X-rays as both the priming and challenge irradiations. The underlying mechanism was due to radio-resistance occurring in blood-forming tissues. In this study, we verified the existence of AR and further investigated residual damage in the hematopoietic system in surviving animals. Results showed that the priming low dose of X-rays could relieve the detrimental effects on the hematopoietic system. We observed both an improvement in the blood platelet count and the ratio of polychromatic erythrocytes (PCEs) to the sum of PCEs and normochromatic erythrocytes (NCEs) and a marked reduction of the incidences of micronucleated PCEs and micronucleated NCEs. These findings suggest that the priming low dose of low linear energy transfer (LET) X-rays induced a protective effect on the hematopoietic system, which may play an important role in both rescue from acute lethal damage (mouse killing) and prevention of late detrimental consequences (residual anhematopoiesis and delayed genotoxic effects) caused by exposure to a high challenge dose from low-LET (X-ray) or high-LET (carbon and neon ion) irradiations. These findings provide new knowledge of the characterization of the Yonezawa Effect by providing new insight into the mechanistic study of AR in vivo.
Figures

Similar articles
-
Adaptive response of low linear energy transfer X-rays for protection against high linear energy transfer accelerated heavy ion-induced teratogenesis.Birth Defects Res B Dev Reprod Toxicol. 2012 Dec;95(6):379-85. doi: 10.1002/bdrb.21027. Epub 2012 Oct 26. Birth Defects Res B Dev Reprod Toxicol. 2012. PMID: 23109298
-
X-ray-induced radioresistance against high-LET radiations from accelerated heavy ions in mice.Radiat Res. 2010 Oct;174(4):532-6. doi: 10.1667/RR2133.1. Radiat Res. 2010. PMID: 20726713
-
Increased Hematopoietic Stem Cells/Hematopoietic Progenitor Cells Measured as Endogenous Spleen Colonies in Radiation-Induced Adaptive Response in Mice (Yonezawa Effect).Dose Response. 2018 Aug 20;16(3):1559325818790152. doi: 10.1177/1559325818790152. eCollection 2018 Jul-Sep. Dose Response. 2018. PMID: 30150909 Free PMC article.
-
Tissue responses to low protracted doses of high LET radiations or photons: early and late damage relevant to radio-protective countermeasures.Adv Space Res. 1989;9(10):299-313. doi: 10.1016/0273-1177(89)90453-5. Adv Space Res. 1989. PMID: 11537307 Review.
-
A review of the bystander effect and its implications for low-dose exposure.Radiat Prot Dosimetry. 2003;104(4):347-55. doi: 10.1093/oxfordjournals.rpd.a006198. Radiat Prot Dosimetry. 2003. PMID: 14579891 Review.
Cited by
-
Immune system modulation by low-dose ionizing radiation-induced adaptive response.Int J Immunopathol Pharmacol. 2023 Jan-Dec;37:3946320231172080. doi: 10.1177/03946320231172080. Int J Immunopathol Pharmacol. 2023. PMID: 37075331 Free PMC article.
-
Altered Response to Total Body Irradiation of C57BL/6-Tg (CAG-EGFP) Mice.Dose Response. 2020 Aug 26;18(3):1559325820951332. doi: 10.1177/1559325820951332. eCollection 2020 Jul-Sep. Dose Response. 2020. PMID: 32922229 Free PMC article.
-
The radiation adaptive response and priming dose influence: the quantification of the Raper-Yonezawa effect and its three-parameter model for postradiation DNA lesions and mutations.Radiat Environ Biophys. 2022 May;61(2):221-239. doi: 10.1007/s00411-022-00963-9. Epub 2022 Feb 12. Radiat Environ Biophys. 2022. PMID: 35150289 Free PMC article.
-
In vivo radioadaptive response: a review of studies relevant to radiation-induced cancer risk.Hum Exp Toxicol. 2015 Mar;34(3):272-83. doi: 10.1177/0960327114537537. Epub 2014 Jun 12. Hum Exp Toxicol. 2015. PMID: 24925363 Free PMC article. Review.
-
Sodium orthovanadate (vanadate), a potent mitigator of radiation-induced damage to the hematopoietic system in mice.J Radiat Res. 2013 Jul 1;54(4):620-9. doi: 10.1093/jrr/rrs140. Epub 2013 Jan 24. J Radiat Res. 2013. PMID: 23349341 Free PMC article.
References
-
- UNSCEAR; UNSCEAR (ed) UNSCEAR 1994 Report to the General Assembly, with Scientific Annexes: Sources and Effects of Ionizing Radiation. Annex B. New York: United Nations: 1994. Adaptive responses to radiation in cells and organisms; pp. 185–272.
-
- Olivieri G, Bodycote J, Wolff S. Adaptive response of human lymphocytes to low concentrations of radioactive thymidine. Science. 1984;223:594–7. - PubMed
-
- Takahashi A, Ohnishi T. Molecular mechanisms involved in adaptive responses to radiation, UV light, and heat. J Radiat Res. 2009;50:385–93. - PubMed
-
- Varès G, Bing W, Tanaka K, et al. Radiation-induced adaptive response with reference to evidence and significance: a review. Indian J Radiat Res. 2006;3:16–34.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

