Cytosolic phospholipase A2 alpha/arachidonic acid signaling mediates depolarization-induced suppression of excitation in the cerebellum
- PMID: 22927908
- PMCID: PMC3425552
- DOI: 10.1371/journal.pone.0041499
Cytosolic phospholipase A2 alpha/arachidonic acid signaling mediates depolarization-induced suppression of excitation in the cerebellum
Erratum in
- PLoS One. 2012;7(11). doi:10.1371/annotation/d08032c8-4a27-4c9c-8ef7-b3ec64351cde
Abstract
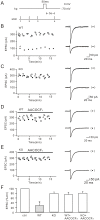
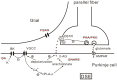
Background: Depolarization-induced suppression of excitation (DSE) at parallel fiber-Purkinje cell synapse is an endocannabinoid-mediated short-term retrograde plasticity. Intracellular Ca(2+) elevation is critical for the endocannabinoid production and DSE. Nevertheless, how elevated Ca(2+) leads to DSE is unclear.
Methodology/principal findings: We utilized cytosolic phospholipase A(2) alpha (cPLA(2)α) knock-out mice and whole-cell patch clamp in cerebellar slices to observed the action of cPLA(2)α/arachidonic acid signaling on DSE at parallel fiber-Purkinje cell synapse. Our data showed that DSE was significantly inhibited in cPLA(2)α knock-out mice, which was rescued by arachidonic acid. The degradation enzyme of 2-arachidonoylglycerol (2-AG), monoacylglycerol lipase (MAGL), blocked DSE, while another catabolism enzyme for N-arachidonoylethanolamine (AEA), fatty acid amide hydrolase (FAAH), did not affect DSE. These results suggested that 2-AG is responsible for DSE in Purkinje cells. Co-application of paxilline reversed the blockade of DSE by internal K(+), indicating that large conductance Ca(2+)-activated potassium channel (BK) is sufficient to inhibit cPLA(2)α/arachidonic acid-mediated DSE. In addition, we showed that the release of 2-AG was independent of soluble NSF attachment protein receptor (SNARE), protein kinase C and protein kinase A.
Conclusions/significance: Our data first showed that cPLA(2)α/arachidonic acid/2-AG signaling pathway mediates DSE at parallel fiber-Purkinje cell synapse.
Conflict of interest statement
Figures




Similar articles
-
Retrograde cPLA2α/arachidonic acid/2-AG signaling is essential for cerebellar depolarization-induced suppression of excitation and long-term potentiation.Cerebellum. 2013 Jun;12(3):297-9. doi: 10.1007/s12311-012-0444-9. Cerebellum. 2013. PMID: 23307660
-
Blockade of 2-arachidonoylglycerol hydrolysis by selective monoacylglycerol lipase inhibitor 4-nitrophenyl 4-(dibenzo[d][1,3]dioxol-5-yl(hydroxy)methyl)piperidine-1-carboxylate (JZL184) Enhances retrograde endocannabinoid signaling.J Pharmacol Exp Ther. 2009 Nov;331(2):591-7. doi: 10.1124/jpet.109.158162. Epub 2009 Aug 7. J Pharmacol Exp Ther. 2009. PMID: 19666749 Free PMC article.
-
Coordinated regulation of endocannabinoid-mediated retrograde synaptic suppression in the cerebellum by neuronal and astrocytic monoacylglycerol lipase.Sci Rep. 2016 Oct 24;6:35829. doi: 10.1038/srep35829. Sci Rep. 2016. PMID: 27775008 Free PMC article.
-
Cerebellar endocannabinoids: retrograde signaling from purkinje cells.Cerebellum. 2015 Jun;14(3):341-53. doi: 10.1007/s12311-014-0629-5. Cerebellum. 2015. PMID: 25520276 Review.
-
Molecules in focus: cytosolic phospholipase A2-alpha.Int J Biochem Cell Biol. 2009 May;41(5):994-7. doi: 10.1016/j.biocel.2008.07.017. Epub 2008 Aug 8. Int J Biochem Cell Biol. 2009. PMID: 18761105 Review.
Cited by
-
Multiple mechanistically distinct modes of endocannabinoid mobilization at central amygdala glutamatergic synapses.Neuron. 2014 Mar 5;81(5):1111-1125. doi: 10.1016/j.neuron.2014.01.012. Neuron. 2014. PMID: 24607231 Free PMC article.
-
Long-term potentiation at cerebellar parallel fiber-Purkinje cell synapses requires presynaptic and postsynaptic signaling cascades.J Neurosci. 2014 Feb 5;34(6):2355-64. doi: 10.1523/JNEUROSCI.4064-13.2014. J Neurosci. 2014. PMID: 24501374 Free PMC article.
-
Numb deficiency in cerebellar Purkinje cells impairs synaptic expression of metabotropic glutamate receptor and motor coordination.Proc Natl Acad Sci U S A. 2015 Dec 15;112(50):15474-9. doi: 10.1073/pnas.1512915112. Epub 2015 Nov 30. Proc Natl Acad Sci U S A. 2015. PMID: 26621723 Free PMC article.
-
Functional compatibility between Purkinje cell axon branches and their target neurons in the cerebellum.Oncotarget. 2017 Aug 1;8(42):72424-72437. doi: 10.18632/oncotarget.19770. eCollection 2017 Sep 22. Oncotarget. 2017. PMID: 29069799 Free PMC article.
-
C-terminal domain of ICA69 interacts with PICK1 and acts on trafficking of PICK1-PKCα complex and cerebellar plasticity.PLoS One. 2013 Dec 16;8(12):e83862. doi: 10.1371/journal.pone.0083862. eCollection 2013. PLoS One. 2013. PMID: 24358315 Free PMC article.
References
-
- Kreitzer AC, Regehr WG (2001) Retrograde inhibition of presynaptic calcium influx by endogenous cannabinoids at excitatory synapses onto Purkinje cells. Neuron 29: 717–727. - PubMed
-
- Maejima T, Ohno-Shosaku T, Kano M (2001) Endogenous cannabinoid as a retrograde messenger from depolarized postsynaptic neurons to presynaptic terminals. Neurosci Res 40: 205–210. - PubMed
-
- Di Marzo V, Fontana A, Cadas H, Schinelli S, Cimino G, et al. (1994) Formation and inactivation of endogenous cannabinoid anandamide in central neurons. Nature 372: 686–691. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

