Cross-priming for antitumor CTL induced by soluble Ag + polyI:C depends on the TICAM-1 pathway in mouse CD11c(+)/CD8α(+) dendritic cells
- PMID: 22934250
- PMCID: PMC3429562
- DOI: 10.4161/onci.19893
Cross-priming for antitumor CTL induced by soluble Ag + polyI:C depends on the TICAM-1 pathway in mouse CD11c(+)/CD8α(+) dendritic cells
Abstract
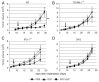
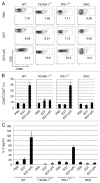
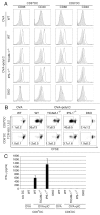
PolyI:C is a nucleotide pattern molecule that induces cross-presentation of foreign Ag in myeloid dendritic cells (DC) and MHC Class I-dependent proliferation of cytotoxic T lymphocytes (CTL). DC (BM or spleen CD8α(+)) have sensors for dsRNA including polyI:C to signal facilitating cross-presentation. Endosomal TLR3 and cytoplasmic RIG-I/MDA5 are reportedly responsible for polyI:C sensing and presumed to deliver signal for cross-presentation via TICAM-1 (TRIF) and IPS-1 (MAVS, Cardif, VISA) adaptors, respectively. In fact, when tumor-associated Ag (TAA) was simultaneously taken up with polyI:C in DC, the DC cross-primed CTL specific to the TAA in a syngenic mouse model. Here we tested which of the TICAM-1 or IPS-1 pathway participate in cross-presentation of tumor-associated soluble Ag and retardation of tumor growth in the setting with a syngeneic tumor implant system, EG7/C57BL6, and exogenously challenged soluble Ag (EG7 lysate) and polyI:C. When EG7 lysate and polyI:C were subcutaneously injected in tumor-bearing mice, EG7 tumor growth retardation was observed in wild-type and to a lesser extent IPS-1(-/-) mice, but not TICAM-1(-/-) mice. IRF-3/7 were essential but IPS-1 and type I IFN were minimally involved in the polyI:C-mediated CTL proliferation. Although both TICAM-1 and IPS-1 contributed to CD86/CD40 upregulation in CD8α(+) DC, H2K(b)-SL8 tetramer and OT-1 proliferation assays indicated that OVA-recognizing CD8 T cells predominantly proliferated in vivo through TICAM-1 and CD8α(+) DC is crucial in ex vivo analysis. Ultimately, tumor regresses > 8 d post polyI:C administration. The results infer that soluble tumor Ag induces tumor growth retardation, i.e., therapeutic potential, if the TICAM-1 signal coincidentally occurs in CD8α(+) DC around the tumor.
Figures



Similar articles
-
Biphasic function of TLR3 adjuvant on tumor and spleen dendritic cells promotes tumor T cell infiltration and regression in a vaccine therapy.Oncoimmunology. 2016 May 19;5(8):e1188244. doi: 10.1080/2162402X.2016.1188244. eCollection 2016 Aug. Oncoimmunology. 2016. PMID: 27622060 Free PMC article.
-
Antitumor NK activation induced by the Toll-like receptor 3-TICAM-1 (TRIF) pathway in myeloid dendritic cells.Proc Natl Acad Sci U S A. 2007 Jan 2;104(1):252-7. doi: 10.1073/pnas.0605978104. Epub 2006 Dec 26. Proc Natl Acad Sci U S A. 2007. PMID: 17190817 Free PMC article.
-
Double-stranded RNA promotes CTL-independent tumor cytolysis mediated by CD11b+Ly6G+ intratumor myeloid cells through the TICAM-1 signaling pathway.Cell Death Differ. 2017 Mar;24(3):385-396. doi: 10.1038/cdd.2016.131. Epub 2016 Nov 11. Cell Death Differ. 2017. PMID: 27834952 Free PMC article.
-
Adjuvant for vaccine immunotherapy of cancer--focusing on Toll-like receptor 2 and 3 agonists for safely enhancing antitumor immunity.Cancer Sci. 2015 Dec;106(12):1659-68. doi: 10.1111/cas.12824. Epub 2015 Nov 18. Cancer Sci. 2015. PMID: 26395101 Free PMC article. Review.
-
Interferon (IFN) and Cellular Immune Response Evoked in RNA-Pattern Sensing During Infection with Hepatitis C Virus (HCV).Sensors (Basel). 2015 Oct 23;15(10):27160-73. doi: 10.3390/s151027160. Sensors (Basel). 2015. PMID: 26512676 Free PMC article. Review.
Cited by
-
The construction of a lymphoma cell-based, DC-targeted vaccine, and its application in lymphoma prevention and cure.Bioact Mater. 2020 Sep 22;6(3):697-711. doi: 10.1016/j.bioactmat.2020.09.002. eCollection 2021 Mar. Bioact Mater. 2020. PMID: 33005832 Free PMC article.
-
Sources of Type I Interferons in Infectious Immunity: Plasmacytoid Dendritic Cells Not Always in the Driver's Seat.Front Immunol. 2019 Apr 12;10:778. doi: 10.3389/fimmu.2019.00778. eCollection 2019. Front Immunol. 2019. PMID: 31031767 Free PMC article. Review.
-
Expression of Toll-like Receptors on the Immune Cells in Patients with Common Variable Immune Deficiency after Different Schemes of Influenza Vaccination.Viruses. 2023 Oct 14;15(10):2091. doi: 10.3390/v15102091. Viruses. 2023. PMID: 37896869 Free PMC article.
-
Toll-like receptor 3 signal augments radiation-induced tumor growth retardation in a murine model.Cancer Sci. 2018 Apr;109(4):956-965. doi: 10.1111/cas.13543. Epub 2018 Mar 25. Cancer Sci. 2018. PMID: 29465830 Free PMC article.
-
Tumoricidal efficacy coincides with CD11c up-regulation in antigen-specific CD8(+) T cells during vaccine immunotherapy.J Exp Clin Cancer Res. 2016 Sep 13;35(1):143. doi: 10.1186/s13046-016-0416-x. J Exp Clin Cancer Res. 2016. PMID: 27619885 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
