Drosophila insulin-like peptide-6 (dilp6) expression from fat body extends lifespan and represses secretion of Drosophila insulin-like peptide-2 from the brain
- PMID: 22935001
- PMCID: PMC3500397
- DOI: 10.1111/acel.12000
Drosophila insulin-like peptide-6 (dilp6) expression from fat body extends lifespan and represses secretion of Drosophila insulin-like peptide-2 from the brain
Abstract
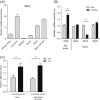
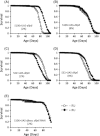
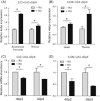
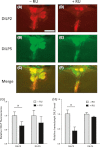
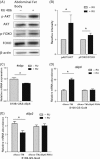
Reduced insulin/IGF signaling extends lifespan in diverse species, including Drosophila melanogaster where the genome encodes seven insulin-like peptides (dilp1-7). Of these, reduced dilp2 expressed in the brain has been associated with longevity assurance when over-expression of dfoxo in fat bodies extends lifespan. Here, we show that the insulin-regulated transcription factor dFOXO positively modulates dilp6 mRNA in adult fat body. Over-expression of dilp6 in adult fat body extends lifespan and increases longevity-associated metabolic phenotypes. Adult fat body dilp6 expression represses dilp2 and dilp5 mRNA in the brain, and the secretion of DILP2 into the hemolymph. The longevity benefit of expressing dfoxo in fat body, and the nonautonomous effect of fat body dfoxo upon brain dilp expression, is blocked by simultaneously repressing dilp6 by RNAi in fat body. dilp6 thus appears to bridge dFOXO, adipose tissue and brain endocrine function to regulate Drosophila longevity.
© 2012 The Authors. Aging Cell © 2012 Blackwell Publishing Ltd/Anatomical Society of Great Britain and Ireland.
Figures

References
-
- Brogiolo W, Stocker H, Ikeya T, Rintelen F, Fernandez R, Hafen E. An evolutionarily conserved function of the Drosophila insulin receptor and insulin-like peptides in growth control. Curr. Biol. 2001;11:213–221. - PubMed
-
- Broughton SJ, Piper MD, Ikeya T, Bass TM, Jacobson J, Driege Y, Martinez P, Hafen E, Withers DJ, Leevers SJ, Partridge L. Longer lifespan, altered metabolism, and stress resistance in Drosophila from ablation of cells making insulin-like ligands. Proc. Natl Acad. Sci. USA. 2005;102:3105–3110. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

