Role of leaky neuronal ryanodine receptors in stress-induced cognitive dysfunction
- PMID: 22939628
- PMCID: PMC3690518
- DOI: 10.1016/j.cell.2012.06.052
Role of leaky neuronal ryanodine receptors in stress-induced cognitive dysfunction
Abstract
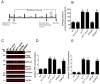
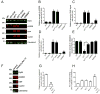
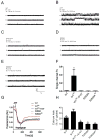
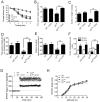
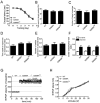
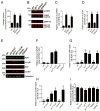
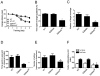
The type 2 ryanodine receptor/calcium release channel (RyR2), required for excitation-contraction coupling in the heart, is abundant in the brain. Chronic stress induces catecholamine biosynthesis and release, stimulating β-adrenergic receptors and activating cAMP signaling pathways in neurons. In a murine chronic restraint stress model, neuronal RyR2 were phosphorylated by protein kinase A (PKA), oxidized, and nitrosylated, resulting in depletion of the stabilizing subunit calstabin2 (FKBP12.6) from the channel complex and intracellular calcium leak. Stress-induced cognitive dysfunction, including deficits in learning and memory, and reduced long-term potentiation (LTP) at the hippocampal CA3-CA1 connection were rescued by oral administration of S107, a compound developed in our laboratory that stabilizes RyR2-calstabin2 interaction, or by genetic ablation of the RyR2 PKA phosphorylation site at serine 2808. Thus, neuronal RyR2 remodeling contributes to stress-induced cognitive dysfunction. Leaky RyR2 could be a therapeutic target for treatment of stress-induced cognitive dysfunction.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
Comment in
-
Receptors: Leaky receptors in stressed brains.Nat Rev Neurosci. 2012 Oct;13(10):668. doi: 10.1038/nrn3356. Nat Rev Neurosci. 2012. PMID: 22992646 No abstract available.
Similar articles
-
Calcium leak through ryanodine receptors leads to atrial fibrillation in 3 mouse models of catecholaminergic polymorphic ventricular tachycardia.Circ Res. 2012 Aug 31;111(6):708-17. doi: 10.1161/CIRCRESAHA.112.273342. Epub 2012 Jul 24. Circ Res. 2012. PMID: 22828895 Free PMC article.
-
Post-translational remodeling of ryanodine receptor induces calcium leak leading to Alzheimer's disease-like pathologies and cognitive deficits.Acta Neuropathol. 2017 Nov;134(5):749-767. doi: 10.1007/s00401-017-1733-7. Epub 2017 Jun 19. Acta Neuropathol. 2017. PMID: 28631094
-
Role of chronic ryanodine receptor phosphorylation in heart failure and β-adrenergic receptor blockade in mice.J Clin Invest. 2010 Dec;120(12):4375-87. doi: 10.1172/JCI37649. Epub 2010 Nov 22. J Clin Invest. 2010. PMID: 21099115 Free PMC article.
-
Calstabin deficiency, ryanodine receptors, and sudden cardiac death.Biochem Biophys Res Commun. 2004 Oct 1;322(4):1267-79. doi: 10.1016/j.bbrc.2004.08.032. Biochem Biophys Res Commun. 2004. PMID: 15336974 Review.
-
Cardiac ryanodine receptor function and regulation in heart disease.Ann N Y Acad Sci. 2004 May;1015:144-59. doi: 10.1196/annals.1302.012. Ann N Y Acad Sci. 2004. PMID: 15201156 Review.
Cited by
-
Sustained Hippocampal Synaptic Pathophysiology Following Single and Repeated Closed-Head Concussive Impacts.Front Cell Neurosci. 2021 Mar 31;15:652721. doi: 10.3389/fncel.2021.652721. eCollection 2021. Front Cell Neurosci. 2021. PMID: 33867941 Free PMC article.
-
Heat shock response and ionstasis: axis against neurodegeneration.Aging (Albany NY). 2012 Dec;4(12):856-8. doi: 10.18632/aging.100517. Aging (Albany NY). 2012. PMID: 23257629 Free PMC article. No abstract available.
-
Regulation of Ryanodine Receptor-Dependent Neurotransmitter Release by AIP, Calstabins, and Presenilins.Adv Neurobiol. 2023;33:287-304. doi: 10.1007/978-3-031-34229-5_11. Adv Neurobiol. 2023. PMID: 37615871
-
The aged rhesus macaque manifests Braak stage III/IV Alzheimer's-like pathology.Alzheimers Dement. 2018 May;14(5):680-691. doi: 10.1016/j.jalz.2017.11.005. Epub 2017 Dec 11. Alzheimers Dement. 2018. PMID: 29241829 Free PMC article.
-
Neurotoxicity and behavioral disorders induced in mice by acute exposure to the diamide insecticide chlorantraniliprole.J Vet Med Sci. 2023 Apr 22;85(4):497-506. doi: 10.1292/jvms.23-0041. Epub 2023 Feb 28. J Vet Med Sci. 2023. PMID: 36858584 Free PMC article.
References
-
- Aguilera G. Regulation of pituitary ACTH secretion during chronic stress. Front Neuroendocrinol. 1994;15:321–350. - PubMed
-
- Airan RD, Meltzer LA, Roy M, Gong Y, Chen H, Deisseroth K. High-speed imaging reveals neurophysiological links to behavior in an animal model of depression. Science. 2007;317:819–823. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

