Structure and interactions of the cytoplasmic domain of the Yersinia type III secretion protein YscD
- PMID: 22942247
- PMCID: PMC3486071
- DOI: 10.1128/JB.00513-12
Structure and interactions of the cytoplasmic domain of the Yersinia type III secretion protein YscD
Abstract
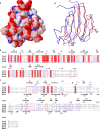
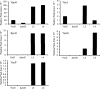
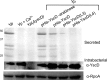
The virulence of a large number of Gram-negative bacterial pathogens depends on the type III secretion (T3S) system, which transports select bacterial proteins into host cells. An essential component of the Yersinia T3S system is YscD, a single-pass inner membrane protein. We report here the 2.52-Å resolution structure of the cytoplasmic domain of YscD, called YscDc. The structure confirms that YscDc consists of a forkhead-associated (FHA) fold, which in many but not all cases specifies binding to phosphothreonine. YscDc, however, lacks the structural properties associated with phosphothreonine binding and thus most likely interacts with partners in a phosphorylation-independent manner. Structural comparison highlighted two loop regions, L3 and L4, as potential sites of interactions. Alanine substitutions at L3 and L4 had no deleterious effects on protein structure or stability but abrogated T3S in a dominant negative manner. To gain insight into the function of L3 and L4, we identified proteins associated with YscD by affinity purification coupled to mass spectrometry. The lipoprotein YscJ was found associated with wild-type YscD, as was the effector YopH. Notably, the L3 and L4 substitution mutants interacted with more YopH than did wild-type YscD. These substitution mutants also interacted with SycH (the specific chaperone for YopH), the putative C-ring component YscQ, and the ruler component YscP, whereas wild-type YscD did not. These results suggest that substitutions in the L3 and L4 loops of YscD disrupted the dissociation of SycH from YopH, leading to the accumulation of a large protein complex that stalled the T3S apparatus.
Figures


Similar articles
-
A C-terminal region of Yersinia pestis YscD binds the outer membrane secretin YscC.J Bacteriol. 2011 May;193(9):2276-89. doi: 10.1128/JB.01137-10. Epub 2011 Feb 25. J Bacteriol. 2011. PMID: 21357482 Free PMC article.
-
Structure of the cytoplasmic domain of Yersinia pestis YscD, an essential component of the type III secretion system.Acta Crystallogr D Biol Crystallogr. 2012 Mar;68(Pt 3):201-9. doi: 10.1107/S0907444911054308. Epub 2012 Feb 7. Acta Crystallogr D Biol Crystallogr. 2012. PMID: 22349221 Free PMC article.
-
Functionally essential interaction between Yersinia YscO and the T3S4 domain of YscP.J Bacteriol. 2013 Oct;195(20):4631-8. doi: 10.1128/JB.00876-13. Epub 2013 Aug 9. J Bacteriol. 2013. PMID: 23935040 Free PMC article.
-
Formation of a Secretion-Competent Protein Complex by a Dynamic Wrap-around Binding Mechanism.J Mol Biol. 2018 Sep 14;430(18 Pt B):3157-3169. doi: 10.1016/j.jmb.2018.07.014. Epub 2018 Aug 2. J Mol Biol. 2018. PMID: 30021116
-
Type III secretion systems shape up as they ship out.Curr Opin Microbiol. 2010 Feb;13(1):47-52. doi: 10.1016/j.mib.2009.11.001. Epub 2009 Dec 16. Curr Opin Microbiol. 2010. PMID: 20015680 Free PMC article. Review.
Cited by
-
A refined model of the prototypical Salmonella SPI-1 T3SS basal body reveals the molecular basis for its assembly.PLoS Pathog. 2013;9(4):e1003307. doi: 10.1371/journal.ppat.1003307. Epub 2013 Apr 25. PLoS Pathog. 2013. PMID: 23633951 Free PMC article.
-
Building a secreting nanomachine: a structural overview of the T3SS.Curr Opin Struct Biol. 2014 Apr;25(100):111-7. doi: 10.1016/j.sbi.2013.11.001. Epub 2014 Apr 1. Curr Opin Struct Biol. 2014. PMID: 24704748 Free PMC article. Review.
-
Identification of Ser/Thr kinase and forkhead associated domains in Mycobacterium ulcerans: characterization of novel association between protein kinase Q and MupFHA.PLoS Negl Trop Dis. 2014 Nov 20;8(11):e3315. doi: 10.1371/journal.pntd.0003315. eCollection 2014 Nov. PLoS Negl Trop Dis. 2014. PMID: 25412098 Free PMC article.
-
The Yersinia pestis type III secretion system: expression, assembly and role in the evasion of host defenses.Immunol Res. 2013 Dec;57(1-3):237-45. doi: 10.1007/s12026-013-8454-3. Immunol Res. 2013. PMID: 24198067 Review.
-
Type Three Secretion System in Attaching and Effacing Pathogens.Front Cell Infect Microbiol. 2016 Oct 21;6:129. doi: 10.3389/fcimb.2016.00129. eCollection 2016. Front Cell Infect Microbiol. 2016. PMID: 27818950 Free PMC article. Review.
References
-
- Akeda Y, Galan JE. 2005. Chaperone release and unfolding of substrates in type III secretion. Nature 437:911–915 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

