High resolution crystal structures of the Cerebratulus lacteus mini-Hb in the unligated and carbomonoxy states
- PMID: 22942687
- PMCID: PMC3430218
- DOI: 10.3390/ijms13078025
High resolution crystal structures of the Cerebratulus lacteus mini-Hb in the unligated and carbomonoxy states
Abstract
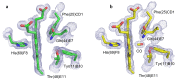
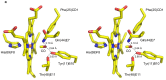
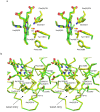
The nerve tissue mini-hemoglobin from Cerebratulus lacteus (CerHb) displays an essential globin fold hosting a protein matrix tunnel held to allow traffic of small ligands to and from the heme. CerHb heme pocket hosts the distal TyrB10/GlnE7 pair, normally linked to low rates of O(2) dissociation and ultra-high O(2) affinity. However, CerHb affinity for O(2) is similar to that of mammalian myoglobins, due to a dynamic equilibrium between high and low affinity states driven by the ability of ThrE11 to orient the TyrB10 OH group relative to the heme ligand. We present here the high resolution crystal structures of CerHb in the unligated and carbomonoxy states. Although CO binds to the heme with an orientation different from the O(2) ligand, the overall binding schemes for CO and O(2) are essentially the same, both ligands being stabilized through a network of hydrogen bonds based on TyrB10, GlnE7, and ThrE11. No dramatic protein structural changes are needed to support binding of the ligands, which can freely reach the heme distal site through the apolar tunnel. A lack of main conformational changes between the heme-unligated and -ligated states grants stability to the folded mini-Hb and is a prerequisite for fast ligand diffusion to/from the heme.
Keywords: carbon monoxide; crystal structure; heme reactivity; nerve globin; protein matrix tunnel.
Figures


Similar articles
-
Two distinct heme distal site states define Cerebratulus lacteus mini-hemoglobin oxygen affinity.Proteins. 2006 Mar 15;62(3):641-8. doi: 10.1002/prot.20822. Proteins. 2006. PMID: 16432879
-
Transient ligand docking sites in Cerebratulus lacteus mini-hemoglobin.Gene. 2007 Aug 15;398(1-2):208-23. doi: 10.1016/j.gene.2007.01.037. Epub 2007 Apr 29. Gene. 2007. PMID: 17531406 Free PMC article.
-
The 109 residue nerve tissue minihemoglobin from Cerebratulus lacteus highlights striking structural plasticity of the alpha-helical globin fold.Structure. 2002 May;10(5):725-35. doi: 10.1016/s0969-2126(02)00763-3. Structure. 2002. PMID: 12015154
-
Nerve globins in invertebrates.IUBMB Life. 2004 Nov-Dec;56(11-12):653-6. doi: 10.1080/15216540500037471. IUBMB Life. 2004. PMID: 15804828 Review.
-
Structural bases for heme binding and diatomic ligand recognition in truncated hemoglobins.J Inorg Biochem. 2005 Jan;99(1):97-109. doi: 10.1016/j.jinorgbio.2004.10.035. J Inorg Biochem. 2005. PMID: 15598494 Review.
Cited by
-
Determination of ligand pathways in globins: apolar tunnels versus polar gates.J Biol Chem. 2012 Sep 28;287(40):33163-78. doi: 10.1074/jbc.M112.392258. Epub 2012 Aug 1. J Biol Chem. 2012. PMID: 22859299 Free PMC article.
References
-
- Bolognesi M., Bordo D., Rizzi M., Tarricone C., Ascenzi P. Nonvertebrate hemoglobins: structural bases for reactivity. Prog. Biophys. Mol. Biol. 1997;68:29–68. - PubMed
-
- Vandergon T.L., Riggs C.K., Gorr T.A., Colacino J.M., Riggs A.F. The mini-hemoglobins in neural and body wall tissue of the nemertean worm, Cerebratulus lacteus. J. Biol. Chem. 1998;273:16998–17011. - PubMed
-
- Pesce A., Nardini M., Dewilde S., Geuens E., Yamauchi K., Ascenzi P., Riggs A.F., Moens L., Bolognesi M. The 109 residue nerve tissue minihemoglobin from Cerebratulus lacteus highlights striking structural plasticity of the α-helical globin fold. Structure. 2002;10:725–735. - PubMed
-
- Pesce A., Nardini M., Ascenzi P., Geuens E., Dewilde S., Moens L., Bolognesi M., Riggs A.F., Hale A., Deng P., et al. ThrE11 regulates O2 affinity in Cerebratulus lacteus mini-hemoglobin. J. Biol. Chem. 2004;279:33662–33672. - PubMed
-
- Martí M.A., Bikiel D.E., Crespo A., Nardini M., Bolognesi M., Estrin D.A. Two distinct heme distal site states define Cerebratulus lacteus mini-hemoglobin oxygen affinity. Proteins Struct. Funct. Bioinforma. 2006;62:641–648. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources

