Fast microbubble dwell-time based ultrasonic molecular imaging approach for quantification and monitoring of angiogenesis in cancer
- PMID: 22943043
- PMCID: PMC3430464
- DOI: 10.3978/j.issn.2223-4292.2012.06.05
Fast microbubble dwell-time based ultrasonic molecular imaging approach for quantification and monitoring of angiogenesis in cancer
Abstract
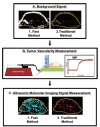
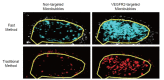
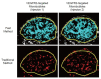
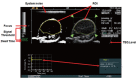
PURPOSE: To develop and test a fast ultrasonic molecular imaging technique for quantification and monitoring of angiogenesis in cancer. MATERIALS AND METHODS: A new software algorithm measuring the dwell time of contrast microbubbles in near real-time (henceforth, fast method) was developed and integrated in a clinical ultrasound system. In vivo quantification and monitoring of tumor angiogenesis during anti-VEGF antibody therapy was performed in human colon cancer xenografts in mice (n=20) using the new fast method following administration of vascular endothelial growth factor receptor 2 (VEGFR2)-targeted contrast microbubbles. Imaging results were compared with a traditional destruction/replenishment approach (henceforth, traditional method) in an intra-animal comparison. RESULTS: There was excellent correlation (R(2)=0.93; P<0.001) between the fast method and the traditional method in terms of VEGFR2-targeted in vivo ultrasonic molecular imaging with significantly higher (P=0.002) imaging signal in colon cancer xenografts using VEGFR2-targeted compared to control non-targeted contrast microbubbles. The new fast method was highly reproducible (ICC=0.87). Following anti-angiogenic therapy, ultrasonic molecular imaging signal decreased by an average of 41±10%, whereas imaging signal increased by an average of 54±8% in non-treated tumors over a 72-hour period. Decreased VEGFR2 expression levels following anti-VEGF therapy were confirmed on ex vivo immunofluorescent staining. CONCLUSIONS: Fast ultrasonic molecular imaging based on dwell time microbubble signal measurements correlates well with the traditional measurement method, and allows reliable in vivo monitoring of anti-angiogenic therapy in human colon cancer xenografts. The improved work-flow afforded by the new quantification approach may facilitate clinical translation of ultrasonic molecular imaging.
Figures



Similar articles
-
Antiangiogenic cancer therapy: monitoring with molecular US and a clinically translatable contrast agent (BR55).Radiology. 2010 Aug;256(2):519-27. doi: 10.1148/radiol.10091858. Epub 2010 Jun 1. Radiology. 2010. PMID: 20515975 Free PMC article.
-
Intra-Animal Comparison between Three-dimensional Molecularly Targeted US and Three-dimensional Dynamic Contrast-enhanced US for Early Antiangiogenic Treatment Assessment in Colon Cancer.Radiology. 2017 Feb;282(2):443-452. doi: 10.1148/radiol.2016160032. Epub 2016 Aug 4. Radiology. 2017. PMID: 27490690 Free PMC article.
-
Three-dimensional ultrasound molecular imaging of angiogenesis in colon cancer using a clinical matrix array ultrasound transducer.Invest Radiol. 2015 May;50(5):322-9. doi: 10.1097/RLI.0000000000000128. Invest Radiol. 2015. PMID: 25575176 Free PMC article.
-
Microbubble-conjugated vascular endothelial growth factor receptor 2 binding peptide.2012 Feb 14 [updated 2012 Jul 11]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2012 Feb 14 [updated 2012 Jul 11]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 22787696 Free Books & Documents. Review.
-
Molecular Ultrasound Imaging.Nanomaterials (Basel). 2020 Sep 28;10(10):1935. doi: 10.3390/nano10101935. Nanomaterials (Basel). 2020. PMID: 32998422 Free PMC article. Review.
Cited by
-
Ultrasound molecular imaging as a non-invasive companion diagnostic for netrin-1 interference therapy in breast cancer.Theranostics. 2018 Oct 6;8(18):5126-5142. doi: 10.7150/thno.27221. eCollection 2018. Theranostics. 2018. PMID: 30429890 Free PMC article.
-
Nanosized Contrast Agents in Ultrasound Molecular Imaging.Front Bioeng Biotechnol. 2021 Nov 29;9:758084. doi: 10.3389/fbioe.2021.758084. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34912789 Free PMC article. Review.
-
In vitro characterization and in vivo ultrasound molecular imaging of nucleolin-targeted microbubbles.Biomaterials. 2017 Feb;118:63-73. doi: 10.1016/j.biomaterials.2016.11.026. Epub 2016 Nov 21. Biomaterials. 2017. PMID: 27940383 Free PMC article.
-
Design considerations for targeted optical contrast agents.Quant Imaging Med Surg. 2012 Dec;2(4):266-73. doi: 10.3978/j.issn.2223-4292.2012.12.04. Quant Imaging Med Surg. 2012. PMID: 23289086 Free PMC article.
-
Dynamic Filtering of Adherent and Non-adherent Microbubble Signals Using Singular Value Thresholding and Normalized Singular Spectrum Area Techniques.Ultrasound Med Biol. 2021 Nov;47(11):3240-3252. doi: 10.1016/j.ultrasmedbio.2021.06.019. Epub 2021 Aug 8. Ultrasound Med Biol. 2021. PMID: 34376299 Free PMC article.
References
-
- Klibanov AL. Microbubble contrast agents: targeted ultrasound imaging and ultrasound-assisted drug-delivery applications. Invest Radiol 2006;41:354-62 - PubMed
-
- Leong-Poi H.Molecular imaging using contrast-enhanced ultrasound: evaluation of angiogenesis and cell therapy. Cardiovasc Res 2009;84:190-200 - PubMed
-
- Hwang M, Lyshchik A, Fleischer AC. Molecular sonography with targeted microbubbles: current investigations and potential applications. Ultrasound Q 2010;26:75-82 - PubMed
-
- Wilson SR, Burns PN. Microbubble-enhanced US in body imaging: what role? Radiology 2010;257:24-39 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
