Cost analysis of HIV treatment and drug-related adverse events when fixed-dose combinations of antiretrovirals (FDCs) were stopped, versus continuation with FDCs
- PMID: 22943676
- PMCID: PMC3484113
- DOI: 10.1186/2191-1991-2-16
Cost analysis of HIV treatment and drug-related adverse events when fixed-dose combinations of antiretrovirals (FDCs) were stopped, versus continuation with FDCs
Abstract
Background: The lower sales price of generic lamivudine has caused healthcare administrators to consider abolishing fixed-dose antiretroviral combinations (FDCs) that contain lamivudine and emtricitabine. The alternative is to administer the individual components of the FDCs separately, thus incorporating the new generic lamivudine medication.
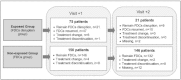
Methods: The Balearic Islands Health Service ordered the discontinuation of the treatment with FDCs in July 2010, but FDCs were reintroduced in August 2010. At that point, an independent, retrospective cost analysis was performed by Son Llàtzer Hospital. A total of 75 patients who were treated from July to August 2010 underwent replacement of their FDC treatment with the individual components. Additionally, 150 patients who continued using FDCs were randomly selected. For both patient groups, the antiretroviral therapy that was administered and the costs associated with management of adverse events were recorded. The study period used for the cost calculations was the average number of days that patients used separate components of FDCs (120 days). An alternative analysis was performed to consider the costs of the extra follow-up visit (consultation and clinical tests) that was required for patients who changed their antiretroviral therapy.
Results: Considering antiretroviral therapies and adverse events, the administration of the separate components increased the total daily cost by 0.72 € per patient compared to treatment with FDCs. When the cost of an extra follow-up visit was considered, the daily cost increased by 3.61 € per patient.
Conclusions: Our study suggests that the discontinuation of FDC treatment and the replacement with the administration of separate antiretroviral agents could lead to an increase in healthcare costs due to the higher rate of adverse events that was observed with the discontinuation of FDCs.
Figures
Similar articles
-
[The analysis of the availability of fixed-dose combinations in antiretroviral therapy for HIV infection in the Russian Federation].Ter Arkh. 2021 Dec 15;93(12):1516-1521. doi: 10.26442/00403660.2021.12.201296. Ter Arkh. 2021. PMID: 36286681 Russian.
-
Legal, ethical, and economic implications of breaking down once-daily fixed-dose antiretroviral combinations into their single components for cost reduction.Enferm Infecc Microbiol Clin. 2014 Nov;32(9):598-602. doi: 10.1016/j.eimc.2013.06.008. Epub 2013 Oct 16. Enferm Infecc Microbiol Clin. 2014. PMID: 24139337
-
Fixed dose combinations of anti-tubercular, antimalarial and antiretroviral medicines on the Indian market: critical analysis of ubiquity, sales and regulatory status.Trop Med Int Health. 2019 Feb;24(2):238-246. doi: 10.1111/tmi.13180. Epub 2018 Nov 26. Trop Med Int Health. 2019. PMID: 30422371
-
Evergreened drugs or evergreened profits?J Eval Clin Pract. 2022 Dec;28(6):1119-1126. doi: 10.1111/jep.13695. Epub 2022 May 11. J Eval Clin Pract. 2022. PMID: 35543377 Review.
-
Fixed-dose combinations for treatment of type 2 diabetes mellitus.Adv Ther. 2012 Jan;29(1):1-13. doi: 10.1007/s12325-011-0094-1. Epub 2012 Jan 16. Adv Ther. 2012. PMID: 22271157 Review.
Cited by
-
A meta-analysis comparing 48-week treatment outcomes of single and multi-tablet antiretroviral regimens for the treatment of people living with HIV.AIDS Res Ther. 2018 Oct 30;15(1):17. doi: 10.1186/s12981-018-0204-0. AIDS Res Ther. 2018. PMID: 30373620 Free PMC article.
-
Past and future of HIV infection. A document based on expert opinion.Rev Esp Quimioter. 2022 Apr;35(2):131-156. doi: 10.37201/req/083.2021. Epub 2022 Jan 12. Rev Esp Quimioter. 2022. PMID: 35018404 Free PMC article. Review.
-
Single-Tablet Regimens in HIV Therapy.Infect Dis Ther. 2014 Jun;3(1):1-17. doi: 10.1007/s40121-014-0024-z. Epub 2014 Feb 20. Infect Dis Ther. 2014. PMID: 25134808 Free PMC article.
-
Meta-Analysis of Studies Comparing Single and Multi-Tablet Fixed Dose Combination HIV Treatment Regimens.Medicine (Baltimore). 2015 Oct;94(42):e1677. doi: 10.1097/MD.0000000000001677. Medicine (Baltimore). 2015. PMID: 26496277 Free PMC article.
-
Effectiveness and tolerability of dolutegravir and abacavir/lamivudine administered as two separate pills compared to their equivalent single-tablet regimen in a multicentre cohort in Spain.J Int AIDS Soc. 2021 Jul;24(7):e25758. doi: 10.1002/jia2.25758. J Int AIDS Soc. 2021. PMID: 34291580 Free PMC article.
References
-
- Epidemiology HIV-1 / AIDS. http://www.unaids.org/en/media/unaids/contentassets/documents/unaidspubl....
-
- Llibre JM, Falco V, Tural C, Negredo E, Pineda JA, Muñoz J, Ortega E, Videla S, Sirera G, Martinez E, Miralles C, Iribarren J, Galindo MJ, Domingo P, D'Arminio-Monforte A, Miro JM, Clotet B. The changing face of HIV/AIDS in treated patients. Curr HIV Res. 2009;7(4):365–377. doi: 10.2174/157016209788680633. - DOI - PubMed
-
- Spanish general council of official colleges of pharmacists - botplus. https://botplusweb.portalfarma.com/. - PubMed
-
- Royal decree-law 4/2010, May 26. http://www.msps.es/profesionales/farmacia/pdf/margenesFactoresConversion....
LinkOut - more resources
Full Text Sources