Toll-like receptor 2 (TLR2), transforming growth factor-β, hyaluronan (HA), and receptor for HA-mediated motility (RHAMM) are required for surfactant protein A-stimulated macrophage chemotaxis
- PMID: 22948158
- PMCID: PMC3481337
- DOI: 10.1074/jbc.M112.360982
Toll-like receptor 2 (TLR2), transforming growth factor-β, hyaluronan (HA), and receptor for HA-mediated motility (RHAMM) are required for surfactant protein A-stimulated macrophage chemotaxis
Abstract
The innate immune system protects the host from bacterial and viral invasion. Surfactant protein A (SPA), a lung-specific collectin, stimulates macrophage chemotaxis. However, the mechanisms regulating this function are unknown. Hyaluronan (HA) and its receptors RHAMM (receptor for HA-mediated motility, CD168) and CD44 also regulate cell migration and inflammation. We therefore examined the role of HA, RHAMM, and CD44 in SPA-stimulated macrophage chemotaxis. Using antibody blockade and murine macrophages, SPA-stimulated macrophage chemotaxis was dependent on TLR2 but not the other SPA receptors examined. Anti-TLR2 blocked SPA-induced production of TGFβ. In turn, TGFβ1-stimulated chemotaxis was inhibited by HA-binding peptide and anti-RHAMM antibody but not anti-TLR2 antibody. Macrophages from TLR2(-/-) mice failed to migrate in response to SPA but responded normally to TGFβ1 and HA, effects that were blocked by anti-RHAMM antibody. Macrophages from WT and CD44(-/-) mice had similar responses to SPA, whereas those from RHAMM(-/-) mice had decreased chemotaxis to SPA, TGFβ1, and HA. In primary macrophages, SPA-stimulated TGFβ production was dependent on TLR2, JNK, and ERK but not p38. Pam3Cys, a specific TLR2 agonist, stimulated phosphorylation of JNK, ERK, and p38, but only JNK and ERK inhibition blocked Pam3Cys-stimulated chemotaxis. We have uncovered a novel pathway for SPA-stimulated macrophage chemotaxis where SPA stimulation via TLR2 drives JNK- and ERK-dependent TGFβ production. TGFβ1, in turn, stimulates macrophage chemotaxis in a RHAMM and HA-dependent manner. These findings are highly relevant to the regulation of innate immune responses by SPA with key roles for specific components of the extracellular matrix.
Figures
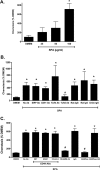
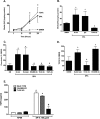
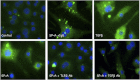
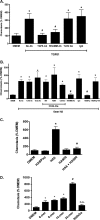




References
-
- Waters P., Vaid M., Kishore U., Madan T. (2009) Lung surfactant proteins A and D as pattern recognition proteins. Adv. Exp. Med. Biol. 653, 74–97 - PubMed
-
- Wright J. R. (2005) Immunoregulatory functions of surfactant proteins. Nat. Rev. Immunol. 5, 58–68 - PubMed
-
- Wright J. R., Youmans D. C. (1993) Pulmonary surfactant protein A stimulates chemotaxis of alveolar macrophage. Am. J. Physiol. 264, L338–L344 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

