β-Actin mRNA compartmentalization enhances focal adhesion stability and directs cell migration
- PMID: 22948660
- PMCID: PMC3435492
- DOI: 10.1101/gad.190413.112
β-Actin mRNA compartmentalization enhances focal adhesion stability and directs cell migration
Abstract
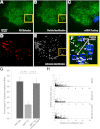
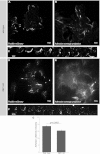
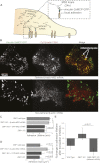
Directed cell motility is at the basis of biological phenomena such as development, wound healing, and metastasis. It has been shown that substrate attachments mediate motility by coupling the cell's cytoskeleton with force generation. However, it has been unclear how the persistence of cell directionality is facilitated. We show that mRNA localization plays an important role in this process, but the mechanism of action is still unknown. In this study, we show that the zipcode-binding protein 1 transports β-actin mRNA to the focal adhesion compartment, where it dwells for minutes, suggesting a means for associating its localization with motility through the formation of stable connections between adhesions and newly synthesized actin filaments. In order to demonstrate this, we developed an approach for assessing the functional consequences of β-actin mRNA and protein localization by tethering the mRNA to a specific location-in this case, the focal adhesion complex. This approach will have a significant impact on cell biology because it is now possible to forcibly direct any mRNA and its cognate protein to specific locations in the cell. This will reveal the importance of localized protein translation on various cellular processes.
Figures

References
-
- Besse F, Ephrussi A 2008. Translational control of localized mRNAs: Restricting protein synthesis in space and time. Nat Rev Mol Cell Biol 9: 971–980 - PubMed
-
- Chan AY, Raft S, Bailly M, Wyckoff JB, Segall JE, Condeelis JS 1998. EGF stimulates an increase in actin nucleation and filament number at the leading edge of the lamellipod in mammary adenocarcinoma cells. J Cell Sci 111: 199–211 - PubMed
-
- Chicurel ME, Singer RH, Meyer CJ, Ingber DE 1998. Integrin binding and mechanical tension induce movement of mRNA and ribosomes to focal adhesions. Nature 392: 730–733 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
