The immunologic functions of the neonatal Fc receptor for IgG
- PMID: 22948741
- PMCID: PMC3548031
- DOI: 10.1007/s10875-012-9768-y
The immunologic functions of the neonatal Fc receptor for IgG
Abstract
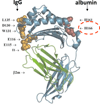
Careful regulation of the body's immunoglobulin G (IgG) and albumin concentrations is necessitated by the importance of their respective functions. As such, the neonatal Fc receptor (FcRn), as a single receptor, is capable of regulating both of these molecules and has become an important focus of investigation. In addition to these essential protection functions, FcRn possesses a number of other functions that are equally as critical and are increasingly coming to attention. During the very first stages of life, FcRn mediates the passive transfer of IgG from mother to offspring both before and after birth. In the adult, FcRn regulates the persistence of both IgG and albumin in the serum as well as the movement of IgG, and any bound cargo, between different compartments of the body via transcytosis across polarized cells. FcRn is also expressed by hematopoietic cells; consistent with this, FcRn regulates MHC class II presentation and MHC class I cross-presentation by dendritic cells. As such, FcRn plays an important role in immune surveillance throughout adult life. The increasing appreciation for FcRn in both homeostatic and pathological conditions is generating an intense interest in the potential for therapeutic modulation of FcRn binding to IgG and albumin.
Conflict of interest statement
Figures


References
-
- Simister NE, Rees AR. Isolation and characterization of an Fc receptor from neonatal rat small intestine. Eur J Immunol. 1985;15(7):733–738. - PubMed
-
- Simister NE, Mostov KE. An Fc receptor structurally related to MHC class I antigens. Nature. 1989;337(6203):184–187. - PubMed
-
- Brambell FW. The transmission of immune globulins from the mother to the foetal and newborn young. Proc Nutr Soc. 1969;28(1):35–41. - PubMed
-
- Brambell FW, Hemmings WA, Morris IG. A Theoretical Model of Gamma-Globulin Catabolism. Nature. 1964;203:1352–1354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- DK51362/DK/NIDDK NIH HHS/United States
- R01 DK088199/DK/NIDDK NIH HHS/United States
- NIH P30DK034854/DK/NIDDK NIH HHS/United States
- R01 DK053056/DK/NIDDK NIH HHS/United States
- AR054407/AR/NIAMS NIH HHS/United States
- P30 DK034854/DK/NIDDK NIH HHS/United States
- NIH AI075037/AI/NIAID NIH HHS/United States
- R21 AR054407/AR/NIAMS NIH HHS/United States
- DK084424/DK/NIDDK NIH HHS/United States
- R37 DK044319/DK/NIDDK NIH HHS/United States
- R01 DK051362/DK/NIDDK NIH HHS/United States
- R01 DK084424/DK/NIDDK NIH HHS/United States
- K08 DK071798/DK/NIDDK NIH HHS/United States
- P30 CA034196/CA/NCI NIH HHS/United States
- NIH DK071798/DK/NIDDK NIH HHS/United States
- DK090603/DK/NIDDK NIH HHS/United States
- R01 DK048106/DK/NIDDK NIH HHS/United States
- R21 DK090603/DK/NIDDK NIH HHS/United States
- R01 AI075037/AI/NIAID NIH HHS/United States
- DK88199/DK/NIDDK NIH HHS/United States
- NIH CA034196/CA/NCI NIH HHS/United States
- R37 DK048106/DK/NIDDK NIH HHS/United States
- NIHDK44319/PHS HHS/United States
- R56 AI075037/AI/NIAID NIH HHS/United States
- R01 DK044319/DK/NIDDK NIH HHS/United States
- DK53056/DK/NIDDK NIH HHS/United States
- R56 DK053056/DK/NIDDK NIH HHS/United States
- NIHDK48106/PHS HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

